Nickel
| Nickel | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General Info | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Symbol | Atomic symbol::Ni | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Number | Atomic number::28 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Weight | Atomic weight::58.69 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical series | Transition Metals | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | lustrous, metallic, silvery tinge 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group, Period, Block | 10, 4, d | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Ar] 4s2 3d8 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2, 8, 16, 2 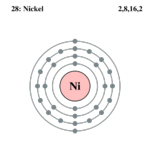
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number | CAS number::7440-02-0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase | solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density | Density::8.9 g/ml | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | Melting point::1726K | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | Boiling point::3005K | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of Nickel | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All properties are for STP unless otherwise stated. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Nickel is a very common chemical element with the symbol Ni. It is the 28th element in the periodic table with an atomic mass of 58.69. Nickel is a transition metal with a silvery appearance. It is the only element named after the devil. Around the 17th century, German miners were trying to remove copper from an ore but could not. They thought that the devil was tricking them so they named the ore "Kupfernickel" which means Old Nick. In fact the element was not copper but nickel. Axel Cronstedt first isolated and discovered nickel. He decided that the name was too long, so he shortened it to the name nickel.[1] Nickel is used almost everywhere from stainless steel to batteries. It is very helpful also and actually beneficial to human health, but only to a limited amount. Over-exposure to nickel can cause serious problems such as respiratory failures and heart disorders. Nickel alloys are used in architecture and tools. Since nickel is abundant in the Earth's crust, it is found almost everywhere, such as in food, water, and air.
Properties
Nickel is a chemical element with the symbol Ni and the atomic number of 28. It was discovered and first isolated in 1751 by Axel Cronstedt in Sweden. The atomic weight is 58.69. Nickel is a transition metal (also known as the iron-cobalt group) in the column VIII B. A physical property of nickel would be that its color is silvery. [2] It is hard but malleable (able to bend). It conducts electricity and heat well. The melting point of nickel is 1453°C while the boiling point is 2732°C. It has the electron configuration of [Ar] 4s2 3d8. Although it is not the only one, the most often used oxidation number of nickel is +2. The other ones are rarely, if not ever, used. [3]
Nickel can also make multiple compounds such as nickel carbonyl which is very toxic. Nickel is one of four elements in the periodic table that are magnetic at room temperature. [4] The density of nickel is 8.90 grams per cubic centimeter. Nickel is somewhat nonreactive. At room temperature, it does not fuse with water or oxygen while at higher temperatures it is more reactive. [1] Nickel does not change at all during chemical reactions so that allows you to use it more than once.[5] Nickel is also resistant to seawater and corrosion.[6]
Occurrences
Nickel occurs with iron, limitedly, in terrestrial deposits. The element makes up 0.007 percent of the Earth's crust. Nickel is mostly condensed in the inner and outer core. Because it is from the Earth's crust, nickel can be found in water, soil, or air. It is twice as plentiful as copper. Nickel is similar to iron in its strength but overall it is more like copper because of its resistance to corrosion. Nickel normally composes igneous rocks which are found abundantly in certain regions of the Earth. [7]
Nickel ranks as the 22nd most abundant element in the Earth's crust. Most of meteorites' composition is made up of nickel. Russia produces the most nickel in the world with one-fifth of the world's production.[8] Nickel can be found naturally in nature but is usually found in ores. It is found minimally in the environment. [6] Nickel is also found in Canadian freshwater and environments because of mining and refining. [9] Food normally contains a small amount of nickel. Chocolate is known to have a high amount of it as well as detergents.[2]
Uses
Nickel is mostly used for the alloys that it makes, such as stainless steel. Stainless steel has many uses such as kitchen appliances and hardware. It is also used for making coins and armor. Other uses include protective coating to other metals. Because of its magnetic ability, Nickel can also be used in magnets, batteries, and ceramics.[3] The nickel itself only contains 25 percent nickel and 75 percent copper. Nickel is used in many everyday products. Surprisingly it is also used in electric guitar strings. Stainless steel is the main product of nickel and it is used in many different things ranging from buildings and architecture to jewelry.[10]
Nickel is used as a catalyst (something that accelerates chemical reaction without affecting itself) for a process called hydrogenation, which is used to make solids of vegetable oils for cooking.[5] Nickel steel can be used for vaults and armor that is very resistant. The compound nickel-cadmium creates rechargeable batteries.[6]
Effect on Humans
Humans can be affected by nickel in many ways from drinking water to smoking cigarettes. Nickel is found almost everywhere usually at small amounts. It is important for humans to intake but only a limited amount because too nickel much is hazardous to human health. Too much exposure to nickel can cause birth defects, heart disorders, and respiratory failures. The effect it has creates serious and life-threatening problems that can stay with humans for their entire life. Nickel in jewelry can cause irritation and at times allergic reactions. Some people may become sensitive to nickel over a period of time and the sensitivity will continue.[2] A large exposure of nickel can cause asthma attacks and lung problems.[11] Vomiting and diarrhea are some effects that over exposure of nickel can have on some people. [12]
References
- ↑ 1.0 1.1 Nickel. Chemistry Explained. Web. Accessed on November 26, 2012. Unknown Author.
- ↑ 2.0 2.1 2.2 Nickel-Ni. Lenntech. Web. Accessed on November 25, 2012. Unknown Author.
- ↑ 3.0 3.1 Helmenstine, Anne Marie Nickel Facts. About. Web. Accessed on November 25, 2012.
- ↑ Nickel. Wikipedia. Web. Last updated on August 22, 2011. Unknown Author.
- ↑ 5.0 5.1 Kavanaugh, John. Nickel Carondelet. Web. Accessed on December 10, 2012.
- ↑ 6.0 6.1 6.2 Nickel Lookchem. Web. Accessed on December 10, 2012. Unknown Author.
- ↑ nickel (Ni). Britannica. Web. Accessed on November 26, 2012. Unknown Author.
- ↑ Nickel. Chemistry Explained. Web. Accessed on November 26, 2012. Unknown Author.
- ↑ Mukherjee, Arun B Occurrences of Nickel in the Canadian environment. Environmental Reviews. Web. Accessed on December 10, 2012.
- ↑ Uses of Nickel. Want to Know it. Web. Accessed on November 26, 2012. Unknown Author.
- ↑ Nickel & compounds: Health effects. National Pollutant Inventory. Web. Accessed on December 10, 2012. Unknown Author.
- ↑ Nickel Compounds. EPA. Web. Last Updated on November 6, 2007. Unknown Author.
| ||||||||||||||
| H | He | ||||||||||||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | ||||||||||
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Uut | Fl | Uup | Lv | Uus | Uuo | ||||||||||
|
|||||||||||||||||||||||||||||||||||||||||


