Barium
| Barium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General Info | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Symbol | Atomic symbol::Ba | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Number | Atomic number::56 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Weight | Atomic weight::137.3 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical series | Alkaline earth metals | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | silvery-white color, metallic luster 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group, Period, Block | 2, 6, s-block | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Xe] 6s2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2, 8, 18, 18, 8, 2 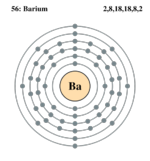
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number | CAS number::7440-39-3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase | Solid at 298K | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density | [[Density::3.5 g.cm-3 at 20°C g/ml]] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | Melting point::725 °C | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | Boiling point::1640 °C | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of Barium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All properties are for STP unless otherwise stated. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
Barium is the 56th element in the periodic table of elements and a member of the alkali earth metals. It has many unique properties that make both itself and it's salts useful to industry. The two electrons in its outermost shell make it react readily. Contact with water, alcohol, or dilute acids cause violent reactions to occur with pure barium. It cannot be found in its pure form in nature like some elements. The element barium has only been identified since 1808, making it relatively new.
Properties
Barium is a soft metallic element with a silvery-white color when pure.[1] It is very malleable and ductile due to how soft it is.[2] It's chemical properties are very similar to Calcium. When stored in open air, Barium quickly oxidizes to form Barium Oxide; to prevent this reaction from occurring the barium must be stored under kerosene or petroleum.[3][4] Barium is a part of the alkaline earth metals, which compose group IIA of the periodic table.[5] Barium has two electrons in its outermost shell making it quite reactive, especially with air; this is why it is not found naturally in its elemental form. It also readily reacts with other non-metals such as the halogens. If in powder form, barium will react violently with air. Other violent reactions can occur if it is combined with dilute acids, alcohol, or water.[6]This metal is malleable like the other elements of group IIA.(Cox, Porch, Wetzel p120)
Occurrences
Barium does not naturally occur in its pure form. Instead, it is usually found combined with a sulfate ion, forming Barium Sulfate (BaSO4). Barium can also be found in other minerals such as Baryte, Witherite, and Barium Carbonate (BaCO3).[7] Barium is primarily mined in the [[United Kingdom, Italy, United States, Czech Republic, and Germany. Barium cannot be found in its pure form so it is usually extracted from baryte and witherite. The annual production of Barium is estimated to be about 6 million tons. It is the 14th most common element in the earth's crust, making up only about 0.0425% of the earth's crust.[8] However, high concentrations of Barium are found only in certain soils and food products.[9]
Uses
Barium is a very useful chemical for several diverse industries. Barium salts such as Barium Nitrate, Ba(NO3)2, and Barium Chlorate, Ba(ClO3)2, give off a green flame when burned, making them very useful for the fireworks.[10]A major application for barium is it's function to remove oxygen from tubes in television sets and other electrons.[11] Drilling through solid rock can be quite difficult, but the oil and gas industries have created a substance called drilling mud to lubricate the drill so it doesn't become stuck. The drilling mud is comprised of many substances including certain Barium compounds. Other products that contain barium compounds are rubber, bricks, glass, tiles, paint, and plastics.[12] The compound Barium Sulfate (BaSO4) is used as a white pigment paint filler but also to add luster to the paint. Barium sulfate, or baryte, has many diverse applications. It is used in the cosmetic industry because of its white hue and safety when applied to the skin. In the rubber industry, baryte is used as a filler to reduce costs while also improving the quality of the rubber. Baryte powder is even used in the paper industry to coat the paper and improve whiteness[13]
History
Barium has a short but interesting history starting in about 1602. An Italian shoemaker named Vincenzo Cascariolo, in the city of Bologna, was experimenting to try and discover the philosopher's stone.[14] He mixed the compound Barium sulphate, commonly called heavy spar at the time, with powdered coal and heated it, then spread it evenly over an iron bar. This gave the iron bar fluorescent properties that could be replenished by the sun. Many were amazed by this new substance and word spread of its amazing qualities. It was called the sun stone at first but later was called the Stone of Bologna after the city it was created in. This was the first mention in history of a barium compound. No one could identify the substances in the Stone of Bologna until one hundred years later, when Swedish chemist Carl Willhelm Scheele discovered the compound baryte. Sir Humphry Davy was the first to isolate barium in 1808 and suggested naming it barium. One chemist suggested naming the new element barytium but barium became the official name of the element.[15]
References
- Barium: the essentials Mark Winter, The University of Sheffield and WebElements Ltd.
- Barium (Ba) Lenntech Water treatment & purification Holding B.V
- Barium Donal O'Leary, 2000.
- 56 Barium Peter van der Krogt, May 5th, 2005.
- BARIUM ChemistryExplained.com, Advameg, Inc.
- Barium Sulfate WWTeq.com, Yi Zhan
- Barium Barium, Wikipedia.org, Multiple authors
- Cox, Heather E., Porch, Thomas E., Wetzel, John S. Chemistry for Christian Schools. Bob Jones University Press.
| ||||||||||||||

