Curium
| Curium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General Info | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Symbol | Atomic symbol::Cm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Number | Atomic number::97 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Weight | Atomic weight::247 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical series | actinide | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group, Period, Block | inner 3,7,F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Rn]5f7, 6d1, 7s2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2,8,18,32,25,9,2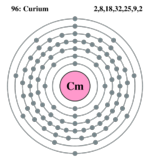
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number | CAS number::7440-51-9 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase | solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density | Density::13.51 g/ml | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | Melting point::1345°C | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | Boiling point::~3100°C | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of Curium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All properties are for STP unless otherwise stated. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The first time curium was produced was in 1944, at the University of California, Berkeley. It was created by Glenn T. Seaborg, Ralph A. James and Albert Ghiorso, but most of the credit is given to Glenn T. Seaborg for the discovery. Curium was named after Marie Curie and Pierre Curie, who discovered polonium and radium. The element was created by neutron capture reactions from the isotopes of plutonium and americium. This happens when plutonium-239 and plutonium isotopes are bombarded with alpha particles that have been accelerated inside a cyclotron. This is how Curium was born, and it product produced isotope Curium 242. Curium 242 has a half-life recorded of about 163 days. Through alpha decay, it will stabilize at plutonium-239. The most stable isotope of curium is isotope-247, which will stabilize back into plutonium-234 through alpha decay. [1]
Properties
Curium is one of the rare radioactive earth metals, and it is part of the transuranic elements on the periodic table. It is so rare that it most likely does not occur in nature, and only can be created nuclear reactor. Since it it is a man made artificial element , the atomic mass standard can not be given. Curium's appearance gives off a silvery metal texture and is a hard, but brittle solid. At room temperature, the metal slowly begins to dry and tarnish. A faint yellow coloration is also seen in most of its compounds.[2]The radioactive properties make curium very dangerous to human health if exposed to it, even compared to some other radioactive elements. Its radioactive properties are so intense that it causes it to glow in the dark.[3]
Link to 3D Curium: crystal structure http://www.webelements.com/curium/crystal_structure_pdb.html
Occurrences
Curium is considered an artificial and man made element, but it possibly does occur in nature. If it truly does occurs in nature by itself, it would be in incredibly small amounts. One theory is that it could be formed in concentrated uranium deposits like plutonium and neptunium. This likely happening is still very low.[4]
It is created by neutron capture reactions from the isotopes of plutonium and americium. This happens when plutonium isotopes are bombarded with alpha particles that have been accelerated inside a cyclotron. Helium ions have also been used in the bombarding process.[5]
Uses
Since Curium is a very radioactive and unstable element, its uses are very limited. It is also limited by it quantity; only a few kilograms are produced each year. Curium has been used in space programs because of its radioactive properties. It is able to produce three watts of heat energy per gram. It has been used in Mars missions for an alpha particle source to be used by the Alpha Proton X-Ray Spectrometer. These space program also require thermic generation for deep space probes, on board satellites, and planetary surface rovers. Curium has been used to power compact thermionic and thermoelectric power generators for a heat sources. It has also been known to be used in pacemakers and in remote navigational buoys.[6]
Glenn T. Seaborg

Curium was discovered by Glenn Seaborg (born in 1912 and died in 1999). He was an American scientist who was widely know for his many discoveries in chemistry for which he won a Nobel prize in 1951. He found and isolated ten elements of what is now part of the actinide series. His findings were plutonium, americium, curium, berkelium, californium, einsteinium, fermium, mendelevium, nobelium and element 106. Even the concept of the actinide series was invented and developed by Seaborg. His finding were so profound that element 106 was named Seaborg in his honor.[7]
References
- ↑ Author-Unknown. [1]. Jefferson Lab. Web. unknown.
- ↑ Author-Winter, Mark . [2]. webelements. Web. copyright 1993-2011.
- ↑ Author-Barbalace, Kenneth . [3]. Environmental Chemistry. Web. Accessed on-line: 11/18/2011.
- ↑ Author-Unknown. [4]. Lenntech. Web. Copyright 1988-2011.
- ↑ Author-Winter, Mark . [5]. webelements. Web. copyright 1993-2011.
- ↑ Author-Unknown . [6]. Royal Society of Chemistry . Web. Accessed on-line: 11/18/2011.
- ↑ Author-Unknown . [7]. The Element Curium. Web. unknown.
| ||||||||||||||
| H | He | ||||||||||||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | ||||||||||
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Uut | Fl | Uup | Lv | Uus | Uuo | ||||||||||
|
|||||||||||||||||||||||||||||||||||||||||

