Calcium
| Calcium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General Info | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Symbol | Atomic symbol::Ca | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Number | Atomic number::20 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Weight | Atomic weight::40.08 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical series | Alkaline earth metal | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | silvery white 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group, Period, Block | 2, 4, s | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Ar] 4s2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2, 8, 8, 2 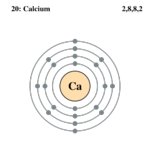
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number | CAS number::7440-70-2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase | solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density | Density::1.55 g/ml | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | Melting point::1115 K | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | Boiling point::1757 K | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of Calcium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All properties are for STP unless otherwise stated. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Calcium is a chemical element known by the chemical symbol Ca. Although very abundant, it can't be found naturally occurring by itself anywhere in our world. Despite this, calcium is very abundant in compounds and is used for many different practical applications. The properties and uses are very unique from many of the other elements on the periodic table. Calcium can be used in a wide variety of substances, ranging from bodily functions, to shell structures of sea-creatures. Without calcium, life on earth would be much harder to maintain.
Properties
Calcium is a relatively solid material that has a silverish tint to its coloring. Calcium is fairly common in the earth and can be found in bones, limestone, and other unique substances. In an element, there are two different categories that are used to distinguish one element from another that might look like it. The two standards used to distinguish an element's traits are physical and chemical properties. Physical properties are those that can be observed by looking at an element without having to use a chemical reaction of sorts to alter it. Chemical properties tell how a chemical will change or react to another element or compound when it comes in contact with it. The physical properties of calcium are: a fairly soft with a cubic crystalline structure and a very high ductility level (this means it can be easily reshaped, or formed into a different thickness/thinness). Calcium is very malleable (can be bent), has a melting point of 1563.8 degrees Fahrenheit and a boiling point of 2699.6 degrees Fahrenheit.[1] Calcium has a specific gravity of 1.55 g/cm and is also an alkaline earth metal. These metals can not be found by themselves, but are often found in compounds.
Other properties of calcium are as follows:
- The fusion heat of calcium is 9.2 Joules per mole
- The evaporation heat of calcium is 153.6 Joules per mole
- The debye temperature of calcium is 230.00 k
- The Pauling Negativity Number of calcium is 1.00
- The lattice structure of calcium is Face-centered cubic
- Calcium has two oxidation states
- Discovered originally by Sir Humphrey Davy
- Atomic radius of 197 picometers
- Covalent radius of 174 picometers[2]
Occurrences
Strangely, calcium can not be found in its free state, but may be found in many different compounds. Calcium can be found on the moon where it constitutes 8% of its crust and on earth where it constitutes a little more than 3.5% of the Earth's crust. As shown in the picture above, calcium can be found in shells of animals, pearls, and in the bones of humans as well. Because of calcium carbonate deposits, stalactites and stalagmites can be formed by the water dripping down, thus forming icicle shaped pillars both coming down and rising up. Calcium can be found in teeth, fluorite, fluorspar, aragonite, gypsum, and other zeolites. Calcium can also be observed in the ocean and salt deposits. In food, calcium is noticeable in milk, sea-creatures(like fish), and certain vegetables as well. Calcium metal has now been created by heating lime with aluminum. Natural calcium consists of mostly the following isotopes: calcium-40, but also contains calcium-44, calcium-42, calcium-48, calcium-43, and calcium-46.[3]
Different compounds of calcium include: Calcium acetate, calcium aluminate, calcium aluminate ferrite, calcium antimonate, calcium arsenate, calcium borate, calcium borohydride, calcium bromide, calcium Carbonate, calcium chlorate, calcium Chloride, calcium chromate(VI), calcium Citrate, calcium, Dietary, calcium disilicide, calcium-eugenol cement, calcium Fluoride, calcium Hydroxide, calcium hypochlorite, calcium lactate, calcium nitrate, calcium Phosphates, calcium selenite, calcium silicate, calcium Sulfate, calcium sulfide, calcium sulfoaluminate, calcium thiosulfate, calcium tungstate, copper hydroxide-calcium, dicalcium silicate, doxaDent, endoBinder, endocal 10, filtra P, GC Altect, green Or, lime, lime-piper betel quid, lime sulfur, mineral trioxide aggregate, perovskite, sandocal, soda lime, tetraaquacalcium perbromate, tobermorite, and tricalcium silicate[4]
Uses
The uses of calcium are very widespread and are needed for many different purposes. These purposes range from making substances, to supporting life on earth. Calcium is probably most well known for being abundant in both human and animal bone. Shells and eggs (like the one shown to the side) need calcium carbonate in order for the shell to harden and serve its purpose to protect and nurture the organism inside of it. Marie Helmenstine said," Calcium is used as a reducing agent when preparing metals from their halogen and oxygen compounds; as a reagent in purification of inert gases; to fix atmospheric nitrogen; as a scavenger and decarbonizer in metallurgy; and for making alloys."(Marie Helmenstine) Calcium can be used to make materials we use in food and building supplies as well. These include: Bricks, concrete, glass, paint products, paper, sugars, and glazes. Calcium is also needed in plants. If there was no calcium in plants, they wouldn't have the ability to grow.[2]
Calcium in the Human Body
In the human body, many different minerals and vitamins are needed in order for the body to function and perform at its maximum capacity. Calcium is a common element in the human body and is necessary for proper functioning of everyday tasks. This is commonly known by most people, but what isn't as commonly known is the fact that calcium is also needed in the central nervous system, to prevent blood clotting, and to help muscles as well. Calcium is normally used in bones, but if your body doesn't get the necessary requirement of calcium for the rest of the body, it will take calcium from the bones in order to continue functioning like it normally would. This may have dire consequences on a person since bones may weaken and become less durable if the person is hit or puts too much strain on it. Osteoporosis can develop if lack of calcium becomes too severe. As of recently, roughly 14 percent of the United states is at risk of developing Osteoporosis(a condition where the bones become brittle or fragile).[5]
Your bones and teeth need calcium in order to become more durable and decrease the risk of breaking. In the central nervous system, calcium actually helps increase brain productivity. Calcium ions help the brain work faster by helping pass signals more efficiently and with a better connection as well. In blood clotting, calcium is indirectly useful to help heal cuts. When you get cut, blood dries and creates a scab around the wounded area so the body can restrict blood flow and heal. The coagulation of blood is permitted and the calcium levels are raised in the body near the infection. This causes reactions that allow blood clots to connect to each other better and improve clotting function. Relatively similar to brain function, calcium helps relay nerve impulses so the body can work more efficiently. The added calcium helps the muscles respond better than it otherwise would have. If you lose too much calcium in your body it may shut down because the heart may stop working or your muscles may twitch due to lack of proper functioning.[5]
References
- ↑ unknown, author. [1] elementalmatter. Web. accessed 11-24-12.
- ↑ 2.0 2.1 Helmenstine, Anne. [2] chemistry.about.com. Web. Accessed 11-24-12.
- ↑ calcium (Ca) Encyclopedia Britannica. Web. Accessed 11-24-12.
- ↑ MDIBL. [3] Comparative Toxicogenomics Database. Web. Updated November 11, 2012.
- ↑ 5.0 5.1 Sherwood, Chris. [4] livestrong. Web. Published May 28, 2011.
| ||||||||||||||
| H | He | ||||||||||||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | ||||||||||
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Uut | Fl | Uup | Lv | Uus | Uuo | ||||||||||
|
|||||||||||||||||||||||||||||||||||||||||


