Gold
| Gold | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General Info | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Symbol | Atomic symbol::Au | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Number | Atomic number::79 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Weight | Atomic weight::196.96655 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical series | Transition Metal | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | soft and golden yellow | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group, Period, Block | group 11, period 6, d-block | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Xe] 4f14 5d10 6s1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
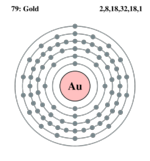
| Electrons per shell | 2, 8, 18, 32, 18, 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number | CAS number::7440-57-5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase | solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density | Density::19.32 g/ml | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | Melting point::1947.9741 °F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | Boiling point::5084.6 °F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of Gold | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All properties are for STP unless otherwise stated. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Gold is one of the most well known and valuable metals. Its atomic symbol is Au, from Latin Aurum, and it belongs to the Transition metals. Gold has been recognized as a rare and precious metal, and has been used for centuries. Its golden yellow glow, and luster make this metal irresistible. Also its malleability, ductility, and excellent conductivity to electricity make gold easy to work with. Because of its qualities, gold has been used in the production of jewelry, coins, electronic devices, and even gold flakes in food.
Gold has been classified as a Precious metal, meaning that it has high economic value, and it also is less reactive than other elements. Gold's lack of reactivity to water and other elements, prevent it from rusting or tarnishing. Gold is also a native mineral, meaning that this element is found as a pure solid in nature. Gold is also mentioned in the Bible multiple times. It was famously used in the making of the Ark of the Covenant, and it also was used as currency. Gold's rarity and usefulness continue to attract people, and its chemical properties allow gold to be one of the most practical metals today.
Properties
Similarly to other metals, Gold is also solid at room temperature. [3]. Gold is also heavy. In the measurement of Density grams per ml, Gold weighs 19.30. It is much heavier than water, which only weighs 1.00. [4]. Gold is also very shiny. Delocalized electrons in gold vibrate in response to light, which leads to them producing their own light. This results in Gold having a lustrous and shiny shimmer. Like most metals, Gold has a very high conductivity. In the Gold lattice, the delocalized electrons move freely and easily, which makes the transfer of electrical current and heat energy very quick. Another property of Gold, is that it is ductile and malleable. Ductility is the property of a material to stretch without being damaged, and malleability is the property of a material to deform under compression. [5]. When only one gram of gold is hammered into a thin sheet, its thickness is about 230 atoms, and it reaches the area of one meter. Also, just one gram of gold can be drawn into a thin wire 541.339 feet long and just 20 micrometers thick. [6].
Occurrences
Gold occurs in many different geological settings,and there are two main types of deposits or occurrences, primary and secondary. In order to produce economic concentrations of gold ore, primary and secondary occurrences both rely on similar physical and chemical processes. In the earth's crust, when chemical reactions between the hydro-thermal (heated water in the earth's crust) mineralizing solutions and rocks occur, gold precipitates. This is where primary deposits form. Secondary deposits form during the chemical and mechanical processed of erosion and weathering.
In most natural substances, there are minor concentrations of gold. In freshwater, there is 0.02 ppb (which is parts per billion). Also in seawater, there is approximately 0.012 ppb of gold. In the earth's crust, the average concentration is 5 ppb. And in some particular sedimentary rocks, there can be concentrations of up to 2100 ppb. In order to extract one ounce of gold, 20 to 30 tons of rock must be processed. [7]
Uses
Gold has many great properties, which makes it very useful and efficient. Gold can be cast and melted into highly detailed shapes, it does not tarnish, it can be molded into wire, it can transform into thin sheets by being hammered down, and it has a beautiful luster and great color. By simply melting the gold nuggets at high temperatures and using molds and hammers, Gold can be made into many shapes and forms. These properties are also reasons why Gold is primarily used in the jewelry industry. Almost 78% of the gold produced each year is in the manufacture of jewelry. [8]
Jewelry: Gold is a precious metal. Precious metals are different from nonmetals because of its value and rarity. Precious metals include platinum, Gold, and silver, while all the other nonmetals fall into the category of Non precious metals.Precious metals are mainly desired for jewelry because it has a higher value, its easy to work with, less reactive than most elements, resistant to tarnish, and they have a higher luster. Often pure gold is mixed with other metals to create white gold, or rose gold. Jewelry made of gold has a level of the how much pure gold it contains, and it is measured by karats.
24 Karat: 99.9% Pure. 22 Karat: 91.7% Pure. 18 Karat: 75% Pure. 14 Karat: 58.3% Pure. 12 Karat: 50% Pure. 10 Karat: 41.7% Pure.
Gold Coins: Gold coins have been used as a currency for centuries. They are still of monumental value today. Many people invest in gold coins, because you can always trust its value, and it could protect you from a stock market crash. The earliest gold coin can be traced back to 6th century BC. They were first produced in Lydia, Western Turkey. They weighed from 17.2 grams to as small as 0.2 grams. On the coin was usually a picture of a lion or a bull, and on the other side was a seal or punch mark. The Romans also used the gold coin, and began printing their emperor's heads on them. Between 200 and 400 AD, millions and millions of gold coins were produced and distributed across the Roman Empire. As gold continued to be discovered, the production of gold coins grew as well. Gold coins reached its highest point after the gold rushes in the United States and Australia after 1848. Still to this day, gold coins have great value. [9].
Other devices: Also because of the effectiveness of Gold, it is been incorporated into some of our daily electronic devices. Small amounts of gold can be found in cell phones, global positioning system units, calculators, and other personal digital assistants. [8]
Biblical accounts of Gold
The King James Bible mentions gold 417 times [10]. Throughout the Bible, Gold is mentioned as a treasure, a currency, as a symbol of wealth and royalty, and used to make false idols or important sculptures and items. In Revelation 21:21, it describes what Heaven will look like. And it describes the streets as Gold. "The twelve gates were twelve pearls, each gate made of a single pearl. The great street of the city was of gold, as pure as transparent glass".
Solomon's temple: This temple was built by Solomon and his workers. It took almost 7 years to complete the temple, and it was solely dedicated to the Lord. In the most holy place of this temple, the room was lined with cedar from Lebanon and it covered with 600 talents of gold. The gold plating by itself, in today's worth would equal to $270 million. Gold plates were also used in the doors of the temple [11] The entire temple used $157,080,000,000 worth of Gold [12].
Building the Ark of the Covenant (Exodus 37). The ark was a rectangular box made of acacia wood, and measured 2 1/2 x 1 1/2 x 1 1/2 cubits (i.e. c. 4 x 2 1/2 x 2 1/2 feet). The whole was covered with gold and was carried on poles inserted in rings at the four lower corners. The lid, or ‘mercy-seat’, was a gold plate surrounded by two parallel placed cherubs with outspread wings. The Ark of the covenant served as a guide to Israel in the wilderness, and serves as a symbol of the presence of God. It was also spoken of as the throne of God. Inside the Ark of the covenant, it contained: the Ten commandments, Aaron's rod, and a pot of manna. It played a crucial part during the fall of Jericho, the ceremony of remembering the covenant at Mt Ebal, and at the crossing of Jordan. [13].
Exodus 37:1-5 "Next Bezalel made the Ark of acacia wood—a sacred chest 45 inches long, 27 inches wide, and 27 inches high. He overlaid it inside and outside with pure gold, and he ran a molding of gold all around it. He cast four gold rings and attached them to its four feet, two rings on each side. Then he made poles from acacia wood and overlaid them with gold. He inserted the poles into the rings at the sides of the Ark to carry it." In Exodus chapter 37, it also refers to the building of the table, the lamp stand, and the incense alter. Gold was incorporated into all of these items. Verse 10: "Then Bezalel made the table of acacia wood, 36 inches long, 18 inches wide, and 27 inches high. He overlaid it with pure gold and ran a gold molding around the edge." Verse 17: "Then Bezalel made the lampstand of pure, hammered gold." This lamp-stand today would cost $1,570,800 for its gold alone. Verses 25-28: "Then Bezalel made the incense altar of acacia wood. It was 18 inches square and 36 inches high, with horns at the corners carved from the same piece of wood as the altar itself.He overlaid the top, sides, and horns of the altar with pure gold, and he ran a gold molding around the entire altar. He made two gold rings and attached them on opposite sides of the altar below the gold molding to hold the carrying poles. He made the poles of acacia wood and overlaid them with gold."
Video
Video of Gold.
References
- ↑ Isotopes of Gold Wikipedia. Web. Last modified October 12, 2016. unknown author.
- ↑ Gagnon, Steve. Isotopes of the element Gold Jefferson Lab. Web. Last accessed November 12, 2016.
- ↑ Batdorf, Brad., Santopietro, Rachel., Cox, Heather., Porch, Thomas., and Wetzel, John. Chemistry. Greenville, S.C: Bob Jones University Press, 2009.page number 123. Print.
- ↑ Ophardt, Charles. Density of Gold Virtual ChemBook. Web.Last accessed November 3, 2016.
- ↑ Kennedy, James. Why is Gold Yellow? James Kennedy. Web. Last accessed November 3, 2016.
- ↑ Winter, Mark. Gold:physical properties WebElements. Web. last accessed November 3, 2016.
- ↑ Weick, James. Gold occurrences Newfoundland Labrador. Web. Last updated July 28, 2015.
- ↑ 8.0 8.1 Puiu, Tibi. How Gold is made and how it got to our planet ZME Science. Web. Published January 12, 2015.
- ↑ . The History of gold coins National Coin Broker. Web. Last accessed November 12, 2016. unknown author.
- ↑ . Bible Investments Bible investments. Web. Last accessed November 12, 2016. unknown author.
- ↑ Dolphin, Lambert. THE TREASURES OF THE HOUSE OF THE LORD Temple Mount. Web. last revised November 1992.
- ↑ . Pulitzer, Hutton. Ark of the Covenant and the ACTUAL Value of the GOLD in King Solomon’s Temple! #GoXplrr Hutton Pulitzer historyheretic. Web. Published November 17, 2014.
- ↑ . What was the Ark of the Covenant, and was it real? Bible.org. Web. Last accessed November 12, 2016. unknown author.
| ||||||||||||||
| H | He | ||||||||||||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | ||||||||||
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Uut | Fl | Uup | Lv | Uus | Uuo | ||||||||||
|
|||||||||||||||||||||||||||||||||||||||||