Titanium
| Titanium | |||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||
| General Info | |||||||||||||||||||||||||||||||||||||||||||||||||
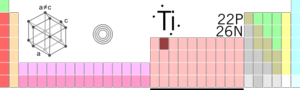
| Atomic Symbol | Atomic symbol::Ti | ||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Number | Atomic number::22 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Weight | Atomic weight::47.867 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical series | Transition metals | ||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | 
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Group, Period, Block | 4, 4, d | ||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Ar] 4s2 3d2 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2, 8, 10, 2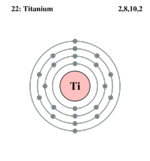
| ||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number | CAS number::7440-32-6 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||||||||||||||||||||
| Phase | solid | ||||||||||||||||||||||||||||||||||||||||||||||||
| Density | Density::4.506 g/ml | ||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | Melting point::1941 K | ||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | Boiling point::3560 K | ||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of Titanium | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| All properties are for STP unless otherwise stated. | |||||||||||||||||||||||||||||||||||||||||||||||||
Titanium is the chemical element known by the atomic symbol Ti. It is best known for its exceptional strength causing it to be used in a great many industrial applications where a strong but lightweight metal is needed.
Properties
Titanium has the atomic number of 22, an atomic mass of 47.867 g/mol and is a solid. It has a melting point of 1941 K and a boiling point of 3560 K. This chemical element also has a density of 4.5 g/cm3. It belongs to period four, group four,[1] and has 8 isotopes. Titanium has a metallic color that is white and silvery[2] as well as being strong enough to withstand up to 200,000 psi.[3] It is also resistant to erosion, lustrous, and lightweight.[3] Titanium has an oxidation number of +4, +3, and +2. Titanium is soluble in concentrated acids but not in water.[2] With two electrons in the third shell and two electrons in the fourth shell, Titanium is known as a transition metal.[3] Possessing a hexagonal structure,[4] Titanium has a low electrical and thermal conductivity as well as being weakly attracted by a magnetic field.[5] It also has a coefficient of linear expansion of about 5.0x10-6 which is low compared to that of copper.[3]
Occurrences
Titanium is very common but hard to extract and because of that pure Titanium (like Titanium Crystal) is very expensive.[6] Titanium is so common that it is in fact the ninth most abundant element in the Earth's crust.[5] The main source of Titanium are the minerals rutile and ilmenite, although it is found in igneous rocks and in sediments that come from those rocks. Titanium has large deposits where it is found in Western Australia, Norway, Canada, Ukraine, North America, and South Africa.[2] Titaniumis also found in the sun and in meteorites.[7] Titanium will always be bound with another element in nature.[8] Titanium has even been found in plants and in humans.[5]
Uses
Titanium is as strong as steel but a lot lighter and because of that it is used in power plants, naval ships, missiles, etc. For medical purposes Titanium is for hip replacements, bone plates, pace-makers, and screws.[2] Titanium is also ideal for bicycle frames, laptops, and jewelry. Titanium is used most as titanium dioxide in plastics, paper, toothpaste, and paints.[8] Titanium dioxide is also used in sunscreen and dental implants. Titanium is found as well in outdoor cookware, construction, and fireworks.[5]
History
Titanium was first discovered by William Gregor in the year 1791.[2] The name Titanium came from the Greek word titanos. William Gregor was a reverend and amateur geologist that found an unknown element in the mineral ilmenite but could not identify it. Later, a German chemist by the name Martin Heinrich Klaproth found it again. He confirmed that it was an unknown element and then named it Titanium in the year 1795.[5]
References
- ↑ Gagnon, Steve. The Element Titanium Jefferson-Lab. Web. 22 January, 2013 date-of-access.
- ↑ 2.0 2.1 2.2 2.3 2.4 Author Unknown. Titanium. Lenntech. Web. 22 January, 2013 date-of-access.
- ↑ 3.0 3.1 3.2 3.3 Author Unknown. Physical Properties of Titanium and Its Alloys. Key to Metals. Web. 22 January, 2013 date-of-access.
- ↑ Bentor, Yinon. Titanium. "Chemical Elements". Web. 22 January, 2013 date-of-access.
- ↑ 5.0 5.1 5.2 5.3 5.4 Author Unknown. All About Titanium. "Berkeley Point". Web. 22 January, 2013 date-of-access.
- ↑ Author Unknown. 22 Ti Titanium. "Images of Elemnets". Web. 08 February 2011.
- ↑ Author Unknown. Titanium The Essentials. "Web Elements". Web. 22 January, 2013.
- ↑ 8.0 8.1 Author Unknown. Titanium Alchemy. "Web-O-Rama". Web. 22 January, 2013.
| ||||||||||||||


