Osmium
| Osmium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General Info | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Symbol | Atomic symbol::Os | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Number | Atomic number::76 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Weight | Atomic weight::190.2 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical series | Transition metals | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | silvery, blue cast 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group, Period, Block | 8B, 6, d | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Xe] 4f14 , 5d-, 6s2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2,8,18,32,14,2 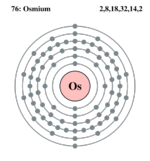
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number | CAS number::7440-04-2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase | Solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density | Density::22.5 g/ml | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | Melting point::3045 °C | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | Boiling point::5027 °C | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of Osmium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All properties are for STP unless otherwise stated. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Osmium is a chemical element classified as a transition metal and known by the chemical symbol Os. It was discovered by an English chemist, Smithson Tennant, in 1803, along with the element Iridium. The name Osmium comes from the Greek word 'osme', meaning the toxic smell, it produces a powdered metal in the air. This powdered Osmium in the air is extremely harmful and it can be deadly to the skin, lung, and eyes. Osmium is the densest of all the elements, meaning it has the highest density, it also has the highest melting point and the lowest boiling point. Osmium is hard, brittle, lustrous, and rare, so it can produce very hard alloys. Osmium is found by melting platinum, and in the mineral osmiridium, an alloy of Osmium and Iridium. Because it can cause serious injuries to people when used incorrectly, Osmium is only allowed to be handled by a chemist. Also, its cost is high because of its rareness. The cost of pure Osmium is $7700 per 100 grams, and bulk Osmium is just a dollar per 100 grams. There are 34 isotopes for this element, and seven of them are naturally formed.[1]
Properties
In the platinum group metals, Osmium is known to have the lowest vapor pressure and the highest melting point. It is lustrous, bluish-white, super hard, and brittle even at high temperatures. The solid structure of Osmium can not be affected by air at room temperature while the powder of osmium can produce Osmium tetroxide, strong oxidizer, highly toxic, with a characteristic odor. It has a melting point of 3045 +/- 30 °C, a boiling point of 5027 +/- 100°C, and is a very hard metal. Its specific gravity is 22.57, with a valence electrons of +3, +4, +6, or +8, and sometimes 0, +1, +2, +5, +7. The color of Osmium is usually blue and white, and it does not melt well even at the high temperatures. Instead, it remains brittle. Osmium is naturally known as the heaviest element with density of 22.61, because it is slightly more dense than Iridium, the second heaviest element. Iridium has the calculated density of 22.65, but when measured it is heavier than osmium. [2] The high melting and boiling points are the highest of any platinum metal, and, moreover, are the highest of all elements. Because of its hardness and inability to melt down and be shaped like most other metals, Osmium is unworkable. Therefore, it has only a few practical uses. After a long period of exposure to liquid, Osmium can be dissolved only by acids and aqua regia. When heated, Osmium can be combined with oxygen, forming Osmium tetroxide(OsO4). Osmium tetroxide is very toxic, and it is the only important commercial compound of Osmium. [3]
Occurrences
With its extreme hardness and resistance, Osmium is usually found bonded to other metals of platinum. The two most well-known ores are Iridosmine and Osmiridium, and they are also the most important ores. It occurs in gold-bearing conglomerates in South Africa, and in gold sands in California and Oregon. It is rare and precious because only less than 100 kilograms are produced per year.[4] [5] The abundance of Osmium is about 0.001 parts per million which makes this element the least abundant element in the Earth's crust. Because of its rareness, the most common ore of Osmium is called osmiridium, and this element occurs in all ores of platinum.[3]
Uses
When Osmium bonds together with other metals in the platinum group, if produce hard alloys, which can make fountain pen tips, instruments, and electrical contacts. OsO4, Osmium tetroxide, can be used to detect fingerprints, and is used in airports to identify people. It is used to stain fatty tissue for microscope slides. [6] Known as a toxic gas, Osmium tetroxide is formed from the powdered and spongy forms of Osmium. Osmium is usually alloyed with platinum and iridium to form tips of various kinds of pens. One platinum Osmium alloy can be used for implants to make pacemakers or replacement valves. [7] Osmium can also be used to increase the hardness of alloys, instrument pivots, and some electrical contacts. [2] Osmium is formed by melting the platinum, and it is sometimes added to platinum and indium to make them harder. Its hardness makes the material of some best pen points, and this alloy is made by melting or mixing two or more metals. Alloys of Osmium and platinum can also be used as materials in specialized laboratory equipment. The other use of Osmium is that it can be used as a catalyst. Catalysts, substances used to adjust the speed of chemical reactions, is used with the finely divided Osmium metal. Since the catalyst does not react by any change during the reaction, it is used for making ammonia from the combination of hydrogen and nitrogen. [3]
Effects
Health Effects
Physical
Because osmium tetroxide is a virulent element, it should only be handled by a chemist. Otherwise it can cause lung congestion, skin damage, and severe eye damage, which is deadly. People can absorb Osmium tetroxide, OsO4, by inhaling its vapor, and by ingestion. At 20°C, this harmful contamination of air evaporates, and it is very dangerous. Once it is inhaled into the body it can cause burning sensations, coughing, headaches, wheezing, trouble breathing, and visual disturbances. When it touches the skin it causes redness, skin burns, skin discoloration, blisters, and pain. When it touches the eyes, it can cause redness, blurred vision, or worse, loss of vision, severe deep burns, and pain. When ingested, it causes abdominal cramps, burning sensation, and shock or collapse.
Chemical
Osmium tetroxide reacts with hydrochloric acid, forming toxic chlorine gas. It also forms unstable compounds with alkalis. It decomposes in heat, and reacts with combustible materials. It causes tears, since it is corrosive to the eyes, the skin, and the respiratory systems. Inhalation of this substance may cause lung oedema(an excessive accumulation of serous fluid in the intercellular spaces of tissue)and exposure to high concentrations may result in death. Repeated or prolonged contact with skin may cause dermatitis and the substance may cause troubles in the kidneys. [4]
History
Osmium was discovered from the element platinum, atomic number of 78, by Smithson Tennant. In the year of 1741, platinum was discovered and, within 60 years, scientists discovered that platinum is actually formed with the mixtures of compounds. In the early 1800s, Smithson Tennant discovered Osmium using aqua regia, a mixture of hydrochloric and nitric acids. He found there is a black powder left out when the element platinum is dissolved in aqua regia. Meaning 'royal water', aqua regia helped to discover Osmium and Indium. After his experiment in 1804, Tennant called the black powder a mixture of two new elements, which are indium and Osmium. The name Osmium came from the unusual smell of Osmium tetroxide. The Greek word 'osme', meaning 'odor', is the foundation of the name of Osmium. [3]
Isotopes
There are seven naturally formed isotopes of Osmium, and the most abundant isotopes are Osmium-192, Osmium-190, and Osmium-189. Respectively, these three isotopes make up 41.0%, 26.4%, and 16.1% of natural Osmium. Isotopes are formed from the alloys of two or more elements, and they differ from each other with their mass number. 192, 190, and 189, numbers on the right side of Osmium, represent the mass number. These mass numbers are the sum of the number of protons and neutrons in the nucleus of an atom. The number of protons represents the element, and number of neutrons can vary in any elements. This variation is an isotope.
Osmium isotopes have many other radioactive isotopes, and a radioactive isotope can break apart to give off form of radiation. This happens when really tiny particles are fired at atoms, and these particles make radioactive stuck in the atoms. But the radioactive isotope of Osmium can be used importantly in any situations. [3]
7 naturally formed isotopes of Osmium
| Isotope | Natural abundance(atom %) | Isotopic enrichment (atom %) |
|---|---|---|
| Os-184 | 0.018 | 5.45 |
| Os-186 | 1.59 | >61 |
| Os-187 | 1.64 | >70 |
| Os-188 | 13.3 | >94 |
| Os-189 | 16.1 | >94 |
| Os-190 | 26.4 | >95 |
| Os-192 | 41.0 | >99 |
Compounds
Osmium forms binary compounds with halogens, oxygen, hydrogen, and some other compounds. A formal oxidation number for Osmium is given for each compound, and it is only limited for p-block elements in particular. The most common oxidation number of known compounds of Osmium is 4.
Fluorides
- Osmium hexafluoride: OsF6: Formula weights 304.22, its color shows yellow on crystalline solid structure. Its melting and boiling points are 32°C and 46°C, respectively. Its density is 4100 kg m-3
- Osmium octafluoride: OsF8:
- Osmium tetrafluoride: OsF4: A crystalline solid, it has yellow color, its melting point is 230°C and formula weight is 266.224.
- Osmium heptafluoride: OsF7: On crystalline structure, it contains yellow color, its melting point is 358°C, with the formula mass of 323.219.
- Osmium pentafluoride: [OsF5]4: With the formula weight of 1140.888, blue color is shown on crystalline structure, which has melting point at 70°C.
Chlorides
- Osmium trichloride: OsCl3: The color dark grey is shown on crystalline solid, which has the melting point at 450°C (decomposes) containing the formula weight of 296.588.
- Osmium tetrachloride: OsCl4: On crystalline solid, it has red color,(black form exists), boiling point at 450°C, density of 4380 kg m-3. It has the formula mass weight of 332.041.
- Osmium pentachloride: OsCl5: With the color of black, where the melting point is at 160°C (decomposes), Osmium pentachloride has formula weight of 367.493.
Bromides
- Osmium tetrabromide: OsBr4: The formula weight is 509.846, black color is shown on solid structure, and it has melting point at 350°C.
Iodis
- Osmium iodide: OsI: With the color of metallic grey, it has the formula weight of 317.134.
- Osmium diiodide: OsI2: 444.039 is Osmium diiodide's formula weight, having black color.
- Osmium triiodide: OsI3: 570.943 of formula weight, this compound also has black color.
Oxides
- Osmium dioxide: OsO2: black or yellow brown colored on crystalline solid, it has density of 11400 kg m-3, and has formula weight at 222.229.
- Osmium tetraoxide: OsO4: pale yellow color is shown on crystalline solid and has 40°C, 130°C; 135°C melting and boiling points respectively. Its density is 5100 kg m-3 at the formula weight of 254.228.
Sulfides
- Osmium disulphide: OsS2: has formula weight of 254.362. Os is 74.79% and S is 25.21% in this compound.
Selenides
- Osmium diselenide: OsSe2: has formula weight of 348.15. Os is 42.71% and Te is 57.29% in this compound.
Tellurides
- Osmium ditelluride: OsTe2: has formula weight of 445.43, and Os has 42.71% and Te has 57.29% in this compound.
Carbonyls
- Dismium nonacarbonyl: Os2(CO)9: The formula weight is 632.551 and orange-yellow color is shown on solid structure. Its melting point is 67°C.
- Triosmium dodecacarbonyl: Os3(CO)12: Yellow color is shown on crystalline solid, with formula weight of 906.81, boiling point at 224°C and its density is 3480 kg m-3.
- Hexaosmium octadecacarbonyl: Os6(CO)18: Dark brown on crystalline solid, its formula weight is 1645.562.
- Octaosmium trieicosacarbonyl: Os8(CO)23: Its formula weight is 2166.072, carbon has 12.75%, oxygen has 16.99%, and osmium has 70.26% amount in this compound.
- Pentaosmium hexadecacarbonyl: Os5(CO)16: the formula weight of this compound is 1399.312, pink-red color is on crystalline solid.
- Oentaosmium nonadecacarbonyl: Os5(CO)19: With the formula weight of 1483.342, orange color is shown on crystalline solid.
- Heptaosmium uneicosacarbonyl: Os7(CO)21: The formula weight of this compound is 1919.822, orange-brown color is shown on a crystalline solid.
Complexes
- Dihydrogen hexabromoosmate: H2[OsBr6]: 671.67 is the formula weight, and it has crystalline structure as well.
- Disodium hexachloroosmiate: Na2[OsCl6]: with 448.926 of formula weight, it is solid.
- Triaquatrichloroosmium: OsCl3(OH2)3:
- Diammonium hexabromoosmiate: (NH4)2[OsBr6]: the formula weight is 705.731, and it is solid.
- Diammonium hexachloroosmiate: (NH4)2[OsCl6]: 439.023 is the formula weight, and it has solid structure with melting point at 170°C, sublimes, and density of 2900.
- Dipotassium hexachloroosate: K2[OsCl6]: The formula weight is 481.143, melting point is decomposed, and it has solid structure.
- Osmocene: Os(C5H5)2: 320.416 is the formula weight, and melting point is around 226-228°C.
- Pentamethylosmocene: Os(C5Me5)2: 460.682 is the formula weight, on solid structure, and it shares carbon of 52.14%, hydrogen of 6.56%, and osmium of 41.29%.
Gallery
References
- ↑ Osmium Element Facts by David D. Hsu. of the Massachusetts Institute of Technology in 1996
- ↑ 2.0 2.1 Osmium Facts-chemical and physical properties by Anne Marie Helmenstine, Ph.D. The New York Times Company, 2010
- ↑ 3.0 3.1 3.2 3.3 3.4 Chemistry Explained Foundation and Applications:Osmium Advameg, Inc. 2007-2010
- ↑ 4.0 4.1 Water Treatment Solutions Lenntech by Alumni from the Technical University of Delft, the Netherlands 1998-2009
- ↑ Science & Technology :: iridosmine Encyclopædia Britannica, Inc., headquartered in Chicago, Illinois 2010
- ↑ Osmium: uses Mark Winter, The University of Sheffield and WebElements Ltd, UK, 1993-2009
- ↑ The Element Collectionby Julianne Gonski 11-26-2010
- ↑ Osmium National Isotope Development Center 1996-2010
- ↑ WebElements: the periodic table on the web by Mark Winter, The University of Sheffield and WebElements Ltd, UK, 1993-2010
| ||||||||||||||





