Radium
| Radium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General Info | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Symbol | Atomic symbol::Ra | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Number | Atomic number::88 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Weight | Atomic weight::226.0254 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical series | Alkaline earth metal | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | silvery white metallic [1].
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group, Period, Block | 2, 7, s [1]. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Rn] 7s2 [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2,8,18,32,18,8,2 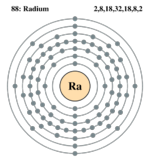
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number | CAS number::7440-14-4 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase | solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density | Density::5.5 g/ml | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | Melting point::700.0 °C | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | Boiling point::1737.0 °C | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of Radium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All properties are for STP unless otherwise stated. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Radium is the 88th element in the periodic table and the heaviest of the alkaline earth metals. Its symbol is Ra and has intense radioactivity. In 1898 radium was discovered by Marie Curie and her husband Pierre Curie. They found this element in pitchblende,massive variety of uraninite, occurring in North Bohemia. Its name derives from the Latin word, radius,which means "ray" [4].
Properties
Radium is a silvery-white when freshly cut, and darkness when exposed to air. It is also a naturally radioactive.Also radium, when purified, glows in the dark. The radiation from radium can make some materials,phosphors, emit light.[5]. It is the heaviest member of the alkaline-earth group and the most explosive. [6]. Metallic radium is extremely chemically reactive, some of the compounds it forms are similar to barium compounds, witch makes the separation of the two elements very difficult.Different isotopes of radium, like Radium-226,-228,and -224, and are made from the radioactive decay of uranium or thorium.[5]
Occurrences
Radium is disseminate in small amounts throughout the earth's crust. It is found in all uranium and thorium minerals. Low concentrations can be found in soil, rocks, surface water, groundwater, plants, and animals. Higher levels of concentration can be found in uranium ores and other geologic materials [7]. Radium was originally found in massive ore in Bohemia. Rich ores of radium are also found in the Great Lake region of Canada and Republic of Zaire. After extensive processing of uranium ores radium is obtained. It is also mined from carnotite sands of Colorado and can be obtained commercially from chlorides and bromides [4] .
Uses
Radium bromide is used to make glow in the dark paint.[6] The paint is used on the dials on watches, clocks, and other instruments [7]. It was also used along with beryllium, as a portable source of neutrons. In the beginning of the 19th century, radium was added to some products like toothpaste, hair creams and even foods. [6]
Medical Uses of Radium
The use of radium in medicine started in the 19th century. Its properties were used to cure different body disorders, like rheumatism and mental disorders. Health hazards where shown after the use of the element and its use was forbidden until research confirmed that it was safe to use. Since radium emits gamma rays, it was used to treat cancer. In 2006, Germany approved the injection of chloride with Radium-224 to treat ankylosing spondylitis, but this treatment is no longer used. The United States has carried out experiments with Radium-224 to produce Bismuth-212 and Lead-212. Melanoma and ovarian cancer are treated with antibodies with those two products. They are capable of destroying cancer cells [4].
Industrial Uses of Radium
Radium is used for the production of Radon gas.Some medical equipment, like calibrators, and lightening rods,are manufacture with Radium-226. A combination of plutonium and radium is used in neutron generators. Since radium undergoes radioactive decay, it can transform to other elements with lower atomic weight, which helps generate other elements. The most common product of radium after disintegration is lead [4].
Health effects of radium
People are always exposed to radium because it is always present in the environment in very small amounts. Humans take a great role in increasing the levels of radium in the environment. People release radium by burning coal and other kinds of fuels. Wells that are located close to a radioactive waste disposal site have higher levels of radium in the water [6] There has been no evidence found to prove that exposure to naturally present levels of radium can be harmful to a person's human health. However, being exposed to high levels of radium can cause some health effects,like teeth fracture,anemia and cataract. When the person has been exposed to radium for a long period of time it may cause cancer or even death. These severe effects are usually caused by gamma radiation of radium and may take years to develop. Radium gamma radiation can travel for long distances through air, so direct contact is not necessary for radium to cause health effects. [6]]
Environmental effects of radium
Radium is produced by the radioactive decay of the elements uranium and thorium. Low levels of Radium can be found in rocks and soil and is strongly attached to these materials. Some can also be found in air.In some locations near uranium mining high levels of radium can be found in the water. Plants close by absorb radium from the soil. Animals that eat these plants consume the radium and accumulate it[6].
References
- ↑ 1.0 1.1 1.2 . Radium Wikipedia. Web. 17 November 2011. author unknown
- ↑ Isotopes of radium. wikipedia. Web. 01 November 2011. author unknown
- ↑ . Basic Atomic Spectroscopic Data. NIST. Web. 29 November 2011.Unknown Author.
- ↑ 4.0 4.1 4.2 4.3 Sengupta,Saptakee. Radium Uses Buzzle. Web. 20 September 2011.
- ↑ 5.0 5.1 Radium. U.S. Environmental Protection Agency. Web. July 08, 2011. Unknown author.
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 Radium - Ra. Lenntech B.V . Web. November 3, 2011. Unknown author.
- ↑ 7.0 7.1 . Radium EVS. Web. August 2005.
| ||||||||||||||
| H | He | ||||||||||||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | ||||||||||
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Uut | Fl | Uup | Lv | Uus | Uuo | ||||||||||
|
|||||||||||||||||||||||||||||||||||||||||


