Tellurium
| Tellurium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|

| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General Info | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Symbol | Atomic symbol::Te | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Number | Atomic number::52 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Weight | Atomic weight::126.9 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical series | Halogens | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | Metallic 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group, Period, Block | 5,5,P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Kr] 4d10, 5s2,5p4 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2,8,18,8,6 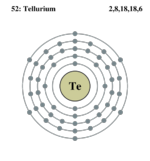
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number | CAS number::13494-80-9 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase | solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density | Density::6.24 g/ml | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | Melting point::449.5°C | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | Boiling point::989.8°C | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of Tellurium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All properties are for STP unless otherwise stated. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Tellurium is a chemical element that has the atomic number 52 and the symbol Te. It is classified as a metalloid and belongs to the same group as sulfur and selenium. Tellurium was discovered in 1782 by Baron Franz Joseph Muller von Reichenstein (1740-1825 or 1826). The element seldom occurs in its pure state, but rather as a compound in ores of gold, silver, copper, lead and mercury.[1]
Properties
Tellurium is a semimetallic, lustrous, crystalline, brittle and silver-white. It is available as a dark grey powder and it has the properties of both the metals and the non metals. Tellurium is formed by many compounds corresponding to those of sulfur and selenium. When burned in air tellurium has a greenish-blue flame and forms tellurium dioxide as a result. Tellurium is unaffected by water or hydrochloric acid, but dissolves in nitric acid.[2]
Tellurium does not dissolve in water. But it does dissolve in most acids and some alkalis. An alkali is a chemical with properties opposite those of an acid. Sodium hydroxide (common lye, such as Drano) and limewater are examples of alkalis.
Tellurium also has the unusual property of combining with gold. Gold normally combines with very few elements. The compound formed between gold and tellurium is called gold telluride (Au 2 Te 3 ). Much of the gold found in the earth occurs in the form of gold telluride.[3]
Occurrences
Tellurium is one of the rarest elements in the Earth's crust. Its abundance is estimated to be about 1 part per billion. That places it about number 75 in abundance of the elements in the earth. It is less common than gold, silver, or platinum.[4] The most common mineral of tellurium is sylvanite. Sylvanite is a complex combination of gold, silver, and tellurium. It is obtained commercially today as a by-product in copper and lead refining. The extreme rarity of tellurium in the Earth's crust is not a reflection of its cosmic abundance, which is in fact greater than that of rubidium, even though rubidium is ten thousand times more abundant in the Earth's crust.[5] There is only a tiny fraction of the metal in Earth's crust that is contained in such rich deposits(Skinner 1976).By far the major fraction of the total amount in the crust is contained in solid solutions in the silicate matrix of common rock.
The supply of metals that are mined as by-products will depend on the mine production of the main product. Tellurium and selenium are by products of copper refining.
Uses
Tellurium used to improve the machining quality of metal products and to color glass and ceramics. Furthermore, Tellurium used in thermoelectric devices, vulcanization of rubber, percussion caps, battery plate protectors and electrical resistors.It can be used for cast iron, ceramics, blasting caps, solar panels, chalcogenide glasses. Furthermore it used in thermoelectric devices, rubber, and battery plate protectors. Tellurium-copper alloys are also easier to work with than pure copper. Tellurium is also added to lead. Tellurium-lead alloys are more resistant to vibration and fatigue than pure lead. Metal fatigue is the tendency of a metal to wear out and eventually break down after long use. [6] Tellurium was used as a chemical binder in the making of the outer shell of the first atom bomb. The 1960s brought growth in thermoelectric applications for Tellurium, as well as its use in free-machining steel, which became the dominant use. Tellurium can be alloyed with some metals to increase their machinability and is a basic ingredient in the manufacture of blasting caps.[7]
References
- http://http://www.chemistryexplained.com/elements/P-T/Tellurium.html,Chemistyexplained
- http://http://www.lenntech.com/periodic/elements/te.htmtitle,lenntech
- http://www.corrosionsource.com/handbook/periodic/52.htm
- http://environmentalchemistry.com/yogi/periodic/Te.html
- http://www.3rd1000.com/elements/Tellurium.htm
| ||||||||||||||
