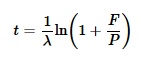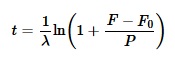Datação radiométrica

A datação radiométrica usa as taxas de decaimento de certos átomos radioativos para datar rochas ou artefatos. Geólogos uniformitaristas consideram essa forma de datação como forte evidência de que a Terra tem bilhões de anos. Mas novas pesquisas feitas pelos criacionistas revelaram um grande número de problemas com a datação radiométrica. Em alguns casos, como na datação por carbono-14, a datação radioativa, na verdade, dá fortes evidências para uma Terra jovem. Outros métodos, como a datação por potássio-argônio, são baseados em suposições incorretas e são tão inconfiáveis que não são úteis.
Princípios básicos
Muitos átomos (ou elementos) existem como numerosas variedades chamadas isótopos, sendo alguns deles radioativos, o que significa que eles decaem ao longo do tempo perdendo partículas. A datação radiométrica é baseada na taxa de decaimento desses isótopos em isótopos não-radioativos, estáveis. Para datar um objeto, os cientistas medem a quantidade de isótopos pai e isótopos filho em uma amostra, e usam a taxa de decaimento nuclear para determinar sua possível idade.
Por exemplo, na série do 238U-206Pb, o 238U é o isótopo pai e os outros são isótopos filho. O 206Pb é o isótopo filho final e o que é analisado na datação radiométrica.
Para calcular a idade da rocha, os geólogos seguem este procedimento:
- Medem a razão dos isótopos na rocha.
- Observam a taxa do decaimento radioativo do isótopo pai para o isótopo filho.
- Calculam o tempo requerido para o isótopo pai produzir todo o isótopo filho observado, de acordo com esta fórmula:
Onde:
- t é a idade do espécime;
- F e P são as quantidades dos isótopos pai e filho atualmente;
- λ é a constante de decaimento para o átomo pai.
No caso especial em que átomos pai e filho estão presentes em igual quantidade, a idade do espécime é a meia-vida do isótopo pai:[1]
Meia-vida (t1/2) é a quantidade de tempo necessária para que a metade dos núcleos em uma amostra radioativa decaia em outro tipo de núcleo.[2]
Suposições
Os vários métodos de datação por radioisótopos dependem de várias suposições básicas. São elas:
- Quantidades conhecidas do isótopo filho (usualmente zero) no início.
- Nenhum ganho ou perda dos isótopos pai ou filho por qualquer outro meio que não o decaimento radioativo (sistema fechado).
- Uma taxa de decaimento constante.[3]
Composição original
A primeira suposição, de que a quantidade do isótopo filho na rocha original é conhecida, é uma suposição muito fraca em alguns casos. Por exemplo, a datação por K-Ar assume que não havia nenhum argônio na rocha original. Mas se havia argônio na rocha quando ela se formou originalmente, então a idade calculada será muito maior do que a idade real.
Para entender isso, relembre a fórmula acima. Quanto maior a quantidade do isótopo filho, maior a idade aparente.
A proporção de argônio para potássio radioativo na amostra hoje é observável, e a constante de decaimento do potássio é prontamente calculável medindo-se a quantidade de argônio produzido a partir do decaimento de 40K após um tempo especificado. Mas a idade da rocha e a proporção de argônio para potássio radioativo na amostra originalmente não são observáveis. Como qualquer estudante de álgebra de primeiro ano logo aprende, uma única equação com duas variáveis desconhecidas não pode ser resolvida. Na verdade, a fórmula acima é simples demais, porque ela assume que a quantidade de isótopo filho era zero no início. A fórmula abaixo é um modelo apropriado que admite a possibilidade de que algum isótopo filho estava presente quando a rocha se formou:
onde F0 é a quantidade de isótopo filho presente no início. Para simplificar a fórmula, os cientistas geralmente assumem que a rocha ígnea não contém nenhum argônio quando se forma, porque o argônio, sendo um gás nobre, iria escapar da lava em esfriamento.
Essa suposição tem sido repetidamente falseada. Rochas vulcânicas recentes são rotineiramente descobertas contendo argônio nelas quando resfriam.[4][5] Nesses casos, lavas de uma idade conhecida de não mais do que alguns milhares de anos (e em um caso, de não mais que dez anos) tinham argônio nelas quando se formaram, de forma que a idade da rocha foi calculada pela datação de K-Ar em milhões de anos, mesmo que fosse conhecido que tinham apenas milhares de anos.
Sistema fechado
Taxa de decaimento
- Artigo principal: Decaimento acelerado
Experimentalmente, a taxa de decaimento (ou meia-vida) de isótopos radioativos não varia significativamente com fatores como temperatura, pressão ou reações químicas, como podemos ler em qualquer livro-texto de física, química ou geologia tratando sobre radioatividade, e os criacionistas que investigam o tópico da datação radiométrica concordam que as taxas de decaimento radioativo são essencialmente constantes no presente. Porém, existem evidências de que as taxas de decaimento radioativo foram alteradas no passado em momentos específicos da história, e também há maneiras teóricas de como isso poderia ter acontecido.
Entre as evidências para o decaimento acelerado dos isótopos radioativos estão: radiohalos de polônio em granito e madeira carbonificada e hélio retido em cristais de zircão. Uma maneira de como as taxas de decaimento poderiam ter sido aceleradas seria através de uma mudança na força nuclear forte.
Os criacionistas acreditam que esse decaimento acelerado ocorreu principalmente durante a criação e durante o dilúvio, com um período de estabilização seguindo o ano do dilúvio.
As evidências para o decaimento acelerado, bem como os mecanismos teóricos para isso, foram investigados pelos físicos e geólogos do grupo RATE. Seus resultados foram documentados principalmente em dois livros técnicos[6][7] e um semi-técnico.[8]
Tipos de Datação Radiométrica
- Datação por carbono-14: Usa a taxa de 14C para 12C para determinar a idade de restos biológicos. Contrariamente à crença popular, a datação por carbono-14 fornece evidência sólida para uma Terra jovem.[9]
- Difusão de hélio: Este método de datação, desenvolvido por criacionistas, é baseado na taxa de difusão de hélio a partir de zircões, que dá a muitas rochas uma idade máxima de 6.000 +/- 2.000 anos.[10]
- Datação por urânio-chumbo
- Datação por potássio-argônio: K-Ar dating was used for a long time despite being challenged by creationists for its faulty assumptions and data. It is no longer defended as reliable, even by uniformitarian geologists, because it is entirely dependent on the assumption that igneous rocks never have any argon when they initially cool, and that assumption has been repeatedly demonstrated to be false as igneous rock of known age has been "dating" to ages far older than its actual age, because there was Argon in it when it formed.[4][5]
- Datação por concórdia
- Isochron dating: Isochron dating was introduced as an attempted substitute for K-Ar dating, after K-Ar's faulty assumptions were exposed. However, isochron dating bears faulty assumptions of its own.
Problemas
Criacionistas têm apontado uma série de problemas tanto com os métodos antigos como com os mais novos de datação radiométrica. Eles citam vários exemplos de datas discordantes quando vários métodos são aplicados à mesma rocha, e várias datações com datas obviamente erradas (incluindo algumas onde a rocha se formou depois de 1900 e foi datada em milhões de anos, como no Mt. Ngauruhoe[11] e no Monte Santa Helena.[4] John Woodmorappe afirma que discrepância de datas é comum, a afirma que cientistas descartam a maioria dos resultados considerados incorretos, dando a ilusão de precisão. Seu livro, The Mythology of Modern Dating Methods, documenta várias citações de geólogos seculares indicando problemas com os vários métodos de datação. Outras questões envolvendo os métodos de datação foram posteriormente investigadas em maior profundidade pelos físicos e geólogos criacionistas do grupo RATE.
Séries de Decaimento Comuns
- Samário-Neodímio. (Sm-Nd)
- Rênio-Ósmio (Re-Os)
- Urânio-Chumbo. (U-Pb)
- Rubídio-Estrôncio (Rb-Sr)
- Potássio-Argônio (K-Ar)
- Lutécio-Háfnio (Lu-Hf)
- Carbono-Nitrogênio (14C-14N)
Referências
- ↑ "Radiometric Time Scale." Em Geologic Time, edição online. USGS Publications Services, 11 de dezembro de 2000. Acessado em 20 de outubro de 2008.
- ↑ Cox, H., Porch, T., Wetzel, J. Chemistry for Christian Schools. Bob Jones University Press; Greenville, South Carolina. (p.533).
- ↑ Morris, JD. "Prologue." Em Radioisotopes and the Age of the Earth, Vardiman L, Snelling AA, e Chaffin EF, eds. El Cajon, CA: Institute for Creation Research, 2000, ISBN 0932766196, p. v.
- ↑ 4,0 4,1 4,2 Austin SA. "Excess Argon within Mineral Concentrates from the New Dacite Lava Dome at Mount St. Helens Volcano." TJ 10(3), 1996.
- ↑ 5,0 5,1 Snelling AA. "'Excess Argon': The 'Achilles' Heel' of Potassium-Argon and Argon-Argon 'Dating' of Volcanic Rocks." ICR Impact 436, janeiro de 1999.
- ↑ Vardiman, L., Snelling, A., e Chaffin, E. (editores), Radioisotopes and the Age of the Earth: A Young-Earth Creationist Research Initiative, Institute for Creation Research e Creation Research Society, 2000.
- ↑ Vardiman, L., Snelling, A., e Chaffin, E. (editores), Radioisotopes and the Age of the Earth: Results of a Young-Earth Creationist Research Initiative, Institute for Creation Research e Creation Research Society, 2005.
- ↑ DeYoung, Don, Thousands... Not Billions: Challenging an Icon of Evolution, Questioning the Age of the Earth. Master Books, 2005.
- ↑ Baumgardner, John, Carbon-14 Evidence for a Recent Global Flood and a Young Earth. Em: Radioisotopes and the Age of the Earth: Results of a Young-Earth Creationist Research Initiative. Institute for Creation Research e Creation Research Society, 2005.
- ↑ Humphreys, DR, Austin, SA, Baumgardner, JR, e Snelling, AA. Helium Diffusion Age of 6,000 Years Supports Accelerated Nuclear Decay. CRSQ 41(1), junho de 2004.
- ↑ Snelling AA. "Andesite Flows at Mt. Ngauruhoe, New Zealand, and the Implications for Potassium Argon 'Dating'." Apresentado na Quarta Conferência Internacional sobre Criacionismo, 1998. Acessado em 20 de outubro de 2008.
Leitura posterior
- Thousands... Not Billions livro de Don DeYoung. 250 páginas em brochura. ISBN 0890514410. 2005
- The Mythology of Modern Dating Methods livro de John Woodmorappe. 118 páginas em brochura. ISBN 978-0932766571. 1999.
Ligações externas
- Basics of Radioactive Isotope Geochemistry Cornell University
- What about carbon dating? The Creation Answers Book, capítulo 4
- A Christian Response to Radiometric Dating por Tas Walker
| |||||||||||
| ||||||||||||||||||||