Protein

Proteins are molecular machines that perform the majority of the biochemical processes within cells. They also serve as the building blocks for many structural components. Their ubiquitous use as builders and building materials alike makes it easy to see why proteins are indeed the material that makes life happen.
Proteins are assembled as part of a complex series of biochemical reactions known as gene expression. Genes are basically the instructions or blueprints that tell the cell how to make proteins. They are found on chromosomes, which are made of DNA. Genes are transcribed by RNA polymerases into messenger RNA (mRNA). Ribosomes then use the mRNA as instructions for linking amino acids together to form a long chain called a polypeptide. After further modification, the polypeptide becomes a protein.
Proteins are composed of twenty different types of amino acids. Amino acids have many structures, but the general formula is NH2-C-COOH. Each contains an amine group (NH2) and a carboxylic acid (COOH). The amino acid residues in a protein are linked together by peptide bonds.
Most proteins form globular structures, but others are chain-like. Proteins have two distinctive types of functions: structural and biological activity. However, some proteins serve both functions. Important structural proteins in animals include collagen, which is the component of the bones, muscles and skin, and keratin, which is the component of the hair, feather, and finger nail.
History
Jons Jakob Berzelius discovered proteins in 1938. He named Protein from the Greek word protas, which means the highest importance.[1]
Function
Proteins have many functions. The most common functions are as structural components of cells and enzymes. Important structural proteins include kertain, actin, myosin, and collagen. Keratin covers skin, hair, and nails.[2] Actin and myosin form muscles.[2] Collagen is a component of ligaments.[2] Enzymes speed up chemical reactions but are not used up by the reaction.
Proteins have other functions as well.
Antibodies
Antibodies are proteins that are part of the immune response. They recognize pathogens by binding to antigens, which are proteins and polysaccharides. This process can either prevent the pathogen from entering cells or mark the pathogen for destruction by phagocytic cells.[3]
Communication
Some hormones are proteins, and some neurotransmitters have protein or amino acid components. Protein hormones are produced by multicellular organisms in one part of the body, are transported to another part of the body, and have an effect in the new location. Neurotransmitters transmit signals between adjacent neurons.
Transportation
Proteins are used to transport molecules from one part of the body to another. In vertebrates, hemoglobin and myoglobin can carry oxygen molecules. Hemoglobin can transport up to four oxygen molecules at one time, but myoglobin can only carry one molecule.
Membrane transport proteins are embedded in plasma membranes and allow the movement of certain molecules across the membrane. They can do this by providing a channel for the molecule to flow according to its concentration or by using energy to actively transport a molecule against its concentration gradient.
Synthesis
- Main article: Protein synthesis
Protein synthesis is the process during which the chemical instructions on genes are used by a living cell to make proteins. It is a highly complex procedure that involves a great number of chemical reactions, both inside and outside the nucleus, which are catalyzed by a host of enzymes and cofactors.
In brief, protein synthesis begins in the nucleus, where the protein-coding instructions from the genes are transmitted indirectly through messenger ribonucleic acid (mRNA), a transient intermediary molecule similar to a single strand of DNA. ThismRNA is moved from the nucleus to the cellular cytoplasm, where it serves as the template for protein synthesis. The cells protein-synthesizing machinery (ribosomes) then translates the codons into a string of amino acids that will eventually constitute the protein molecule.[4]
| Examples of nucleotide codons and their corresponding amino acid: Codon - Amino Acid TGC = Cysteine CTG = Leucine AGT = Serine GCA = Alanine |
Structure
The product of a single gene is a string of amino acids known as a peptide or polypeptide. This is known as the primary structure of a protein. This peptide is folded into a particular subunit, and then joined together with other peptides to make a functional protein. Protein structures are known by 4 levels of organization: primary, secondary, tertiary, and quaternary protein structures.[5]
Primary
The first structure of protein is primary protein structure or primary protein stage. The primary structure consists the repeating sequence of three atoms: N-C-C. N is alpha helix, and C is carboxyl group. The structure of proteins is bonded by the covalent bonds. From the primary stage, the translation is occurring; the amino acids form to bond into the special order of them. (Polypeptide, group of amino acids, is the sequence of amino acids in the primary stage.) Proteins can be distinguished by the stable structures.[6]
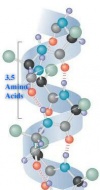 | 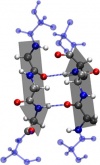 |
Secondary
Secondary structure is occurring after the primary structure. It consists of the regular repeating patterns in the polypeptide chains. Hydrogen bonding plays an important role in stabilizing these folding patterns. In the secondary structures, there are two different types of the structures: alpha helix and beta pleated sheet. Each of the structures work differently, even though they are occurring in the secondary structure.[6] The formation of these structures are determined by the primary structure.
- Alpha helix: Alpha is a right handed coil. The alpha helical secondary structure is common in keratins, which is the fibrous structural protein: hair, feather, and toe nails.
- Beta pleated sheet: Beta pleated sheet is formed from two or more polypeptide chains. It is stabilized by the hydrogen bonds between N-H and C-O groups.
Tertiary
Of course, tertiary structure occurs after the secondary structure. The tertiary structure is the polypeptide and proteins are composing it as the secondary structure of protein. Polypeptides in tertiary structure can be bent and be folded back and forth. Many of the interactions between atoms are occurred in tertiary structure.[6]
- Covalent disulfide bridges: They can hold the folded polypeptides
- Hydrophobic: Side chains are gathered together in protein to fold the polypeptides that the process occurs away from water
- Van der Waals force: It stabilize the interactions between the hydrophobic residues.
- Ionic bonds: They form between positive and negative charges.
Quaternary
The quaternary structure describes the interaction of proteinaceous subunits with other subunits or different molecules. The quaternary stricture is specifically the result of binding of the subunits together and their interactions. The protein subunits are the short tertiary polypeptide chains. The tetrameric metalloprotein hemoglobin is a good illustration of the quaternary structure. Hydrogen bonds, ionic bonds, Van der Waals forces, and hydrophobic bonds hold four different subunits together (2 α subunits and 2β subunits) along with an Fe(II) ion. The presence of the iron allows oxygen binding and transportation in the form of oxyhemoglobin during cellular respiration. When oxyhemoglobin releases O2 in the cells of the body, the quaternary structure undergoes a change.[6]
Protein
References
- ↑ Some Protein History. Associated Content.
- ↑ 2.0 2.1 2.2 Bender, Hal. Structural Proteins. Clackamas Community College. 2003. Accessed 20 May 2016.
- ↑ Janeway CA Jr, Travers P, Walport M, et al. The Humoral Immune Response. In Immunobiology: The Immune System in Health and Disease. 5th edition. New York: Garland Science; 2001.
- ↑ Primer on Molecular Genetics: Introduction by the U.S. Department of Energy, Accessed January 22, 2011.
- ↑ Protein and Polypeptide Structure: Four Conformation Levels of Protein Structure By Anne Marie Helmenstine, Ph.D., About.com Guide, Accessed January 22, 2011.
- ↑ 6.0 6.1 6.2 6.3 Mark McDowall and Neil Cameron. Protein: Primary Structure. Chemsoc. The Royal Society of Chemistry.
- Protein & Polypeptide Structure. About Chemistry.
| ||||||||||||||
| |||||||||||||||||






