Electrochemical cell
The electrochemical cell is the main electric current that supplies batteries with their power. This cell is a device that is capable of either generating either electrical energy from chemical reactions, or facilitating chemical reactions through the introduction of electrical energy. The electrochemical cell consists of two main parts, which are the electrolytic cell and the galvanic cell. The electrolytic cell is a cell that causes nonspontaneous reactions which modify electrical energy into chemical energy. The galvanic cell is a cell that causes spontaneous reactions which modify chemical energy into electrical energy. A name given to these reactions that occur within the cells is redox reactions. Redox reactions also consist of two main processes which are known as reduction and oxidation. Because of the reactions that take place in an electrochemical cell, they are used in a wide range of differing kinds of batteries. Some of these batteries include primary batteries and lead-acid storage batteries.
Electrolytic cell
An electrolytic cell is a cell that exudes chemical energy through a method known as electrolysis. Electrolysis is the process in which an electric current flows through a mixture in order to create a non-spontaneous reaction (a reaction that is induced by external sources). The main function of the electrolytic cells is to convert electrical energy into chemical energy. Because the reactions in an electrolytic cell are non-spontaneous, an outside supply of electrical energy is required in order for the cell to complete this function. This supply of electrical energy causes a redox reaction to take place which, in turn, transforms the electrical energy into chemical energy and thus completing the process.[1] Redox reactions play a major role in both electrolytic cells and in galvanic cells. In electrolysis they are the main reaction responsible for the conversion of electrical energy into chemical energy. A redox reaction can be defined as a reaction in which the electrons in one compound transfer to another and the oxidation number of that compound is changed. A redox reaction also consists of two main processes. These processes are oxidation and reduction. During the reaction, the compound or single element that loses electrons is said to be oxidized, while the compound or element that gains electrons is said to be reduced. Therefore, when the charge of a element becomes more positive, it is being oxidized and when the charge becomes more negative, it is being reduced. Some abbreviations commonly used for these two processes are LEO (loss equals oxidation) and GER (gain equals reduction). Oxidation and Reduction always occur simultaneously during a redox reaction.[2]
[3] Similarly to galvanic cells, the electrolytic cell consists of several parts that make up its structure. These parts are referred to as the anode, the cathode, and the salt bridge. The anode is positively charged and reacts through oxidation. The cathode is negatively charged and reacts through reduction. Electrons flow from the cathode to the anode in electrolytic cells. The job of the salt bridge is to carry out the circuit as well as to the two half cells together. Metals, such as sodium, and gases, such as chlorine, are often formed from electrolytic cells. Sodium ions convert to metal at the cathode through the process of reduction, and chlorine ions convert to gas at the anode through the process of oxidation.[4] Electrolysis is most often seen in batteries that are rechargeable and the process takes place during the charging of the battery.[5] Electrolytic cells have a much higher voltage than that of the galvanic cell. This is because enough voltage is created simply from the circuit. The stronger voltage is required in order to push the flow of the electrons in the opposite direction that they would naturally flow, as they do in the galvanic cells. [6] In order to balance this flow of electrons out, anions (negatively charged ions) must migrate towards the cathode and cations( positively charged ions) must migrate towards the anode.
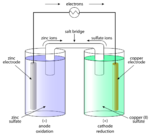
Galvanic cell
Galvanic cells, also known by the name of voltaic cells, are electrochemical cells that generate electric currents. Opposite of electrolytic cells, the main function of galvanic cells is to convert chemical energy into electrical energy. This conversion takes place through spontaneous reactions. In other words, no external forces are needed to produce the reaction.[7] Galvanic cells are responsible for the electric currents in batteries. A battery is made up of a pair of connected galvanic cells. The reactions that take place in galvanic cells give batteries their power by exuding electricity.
Galvanic cells consist of several individual structures, all of which work in unison to makes up the entire cell. Similarly to electrolytic cells, galvanic cells possess an anode, cathode, and a salt bridge. The anode and cathode are the names for two structures called electrodes. These two electrodes are connected by an electrically charged device called an electrolyte. The electrolyte is the passage, containing ions, that allows electrons to migrate from one electrode to the other. As it is opposite of the electrolytic cell, electrons in the galvanic cell flow from the anode to the cathode.[8] Other structures making up the galvanic cell include the external circuit and the internal circuit.[9] The function of the salt bridge is to also serve as a connecting point. It joins the two half cells, where each half of the redox reaction occurs (Oxidation at one half and Reduction at the other) in order to create a neutral electrical charge inside of the internal circuit.[10] The external circuit is the pathway for electrons to move between electrodes, while the internal circuit is a pathway for particles that are charged.[11] The process galvanic cells use takes place during the use of batteries.
Batteries
Batteries come in variety of forms and sizes. They range from batteries that are used in cars to those that are required in order to power a remote control or a cellular device. Electrochemical cells are the source components that make up batteries.
In lead-acid storage batteries, both galvanic and electrolytic cells are used to create electric currents. The cathode contains lead dioxide discs and the anode contains porous lead discs, both of which are combined with a mixture of sulfuric acid. A reaction between lead and sulfate ions create lead sulfate. This reaction occurs during the time the battery is charging and discharging.[12]
Because primary batteries are dispensable and cheap compared to lead-acid batteries, they are often seen used in utilities such as flashlights or remote controls. In primary batteries, the anode, or the outside of the battery, is formed from Zinc. The zinc is oxidized, which creates a reaction in the anode. Manganese dioxide is reduced, creating a reaction in the cathode. Through processes which includes electrolytic and galvanic cells, electrons flow back and forth through a circuit, giving the battery function. When the electrons in the circuit cease the continuous of back and forth, the battery dies and must be replaced.[13]
References
- ↑ Spontaneous and Nonspontaneous Reactions ck12.org. Published June 25, 2013. Unknown Author.
- ↑ What are Electrolytic and Galvanic Cells? CheggTutors. Acessed June 6, 2017. Unknown Author.
- ↑ ELECTROLYTIC CELL VS GALVANIC CELL Chemistry Blogspot. Published February 15, 2011. Unknown Author.
- ↑ Helmenstine, Anne Marie.Electrochemical cells About Education. Accessed November 12, 2016.
- ↑ Cell Conventions Chemistry LibreTexts. Last-modified April 20, 2014. Unknown Author.
- ↑ ELECTROLYTIC CELL VS GALVANIC CELL Chemistry Blogspot. Published February 15, 2011. Unknown Author.
- ↑ Galvanic Cell Corrosionpedia. Accessed November 2, 2016. Unknown Author.
- ↑ A voltaic cell is a device that produces an electric current from energy released by a spontaneous redox reaction in two half-cells Boundless.com. Accessed June 6, 2017. Unknown Author.
- ↑ Electrochemistry: Voltaic Cells ChemPages Netorials. Accessed November 3, 2016. Unknown Author.
- ↑ Chemistry of Batteries uwaterloo.ca. Accessed November 8, 2016. Unknown author.
- ↑ Maqsudi, Murtaza. Galvanic Cell biochemreview.weebly.com. Accessed November 13, 2016. Month Day, Year.
- ↑ Zoski, Cynthia G. electrochemistry chemistry Explained. Accessed November 1, 2016.
- ↑ Woodford, Chris. Batteries Explainthatstuff. Last-modified August 20, 2016.
| ||||||||||||||