Ammonia
| Ammonia | |
|---|---|
| |
| General | |
| Systematic name | Ammonia, Azane |
| Other names | Hydrogen nitride Spirit of hartshorn |
| Molecular formula | NH3 |
| Molar mass | Molar mass::17.0304 g/mol |
| Appearance | colorless gas with
strong pungent odor |
| CAS number | CAS number::7664-41-7 |
| Properties | |
| Density and phase | [[Density::0.6813 g/cm3]], gas |
| Solubility in water | 89.9 g/100 ml (0°C) |
| Melting point | Melting point::-77.73°C (195.42 K) |
| Boiling point | Boiling point::-33.34°C (239.81 K) |
| Acidity (pKa) | 33.6 |
| Basicity (pKb) | 4.75 |
| Structure | |
| Dipole moment | 1.42 D |
| Molecular shape | Terminus |
| Hazards | |
| MSDS | External MSDS |
| Main hazards | Toxic and Corrosive |
| NFPA 704 | |
| R/S statement | R: R10, R23, R24, R50 S: S1/2, S16, S36/37/39, |
| RTECS number | BO0875000 |
| Related compounds | |
| Other anions | Ammonium (NH4+) |
| Related compounds | Hydrazine Hydrazoic acid Hydroxylamine Chloramine |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Disclaimer and references | |
Ammonia, or NH 3, usually exists in a gaseous state, and can be acquired fairly easy. This ammonia gas has a very pungent and distinct odor. Ammonia is the definition of the "Good, Bad and Ugly", meaning it contributes in so many good ways, but the gas alone is very toxic and corrosive. Ammonia is colorless and is lighter than air.
Ammonia has very many types of uses such as: nitric acid production, an universal indicator, fertilizer, refrigeration, disinfectant and fuel. These some of the many uses ammonia has to offer.
Ammonia is used commercially, but most people just know it as a typical household cleaner
Properties
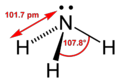
The molecular shape of ammonia is trigonal planar, meaning the bond angle existing between each nitrogen and hydrogen bond is set at, roughly, 108 degrees. There is one existing nitrogen atom in a single molecule of ammonia. Nitrogen has a single bond between each evenly spaced hydrogen atom. The molecular shape highly determines what properties ammonia will have. The shape of ammonia makes this particular molecule polar and allows it to easily dissolve in water. A typical ammonia molecule exists in a gaseous state, that is colorless, with a recognizable, odoress smell. Ammonia's density is roughly half of air's density causing it to be lighter than air. Liquid ammonia boils at -33.3 degrees Celsius and solidifies at -77.7 degrees Celsius. Ammonia is miscible in water and can easily be expelled from water and exist in a gas state. Ammonia's density when saturated in water is .88 g/mol, and widely known as ".880 Ammonia."
Ammonia does not readily combust, unless combined with oxygen, expelling a greenish-yellow flame. When in the presence of a suitable catalyst and at fairly high temperatures, ammonia will lose its bonds and separate.[1]
History
Ammonia salts can be dated back to earlier times, this is true because we see the term, "Hammoniacus sal", scribbled down in writings of the Pliny. Sal-ammoniac was officially recognized by alchemists due to Albertus Magnus in the 13 th century. Ammonia was also used as a form of fermented urine by dyers to help differ the color in vegetable dyes. By the 15 th century, Basilius Valentinus proved a different and easier way to obtain sal-ammoniac through a process involving alkalis. Before discovering this easier method, sal-ammoniac was created through process of distilling hoofs of oxen, thus the name of "Spirit of the Hartshorn."
Joseph Priestley was the first to isolate ammonia in a gaseous state but Basil Valentine was the man you collected and contained it. 1785, Claude Louis Berthollet was the person to confirm what it was made out of.[2]
Health
Ammonia occurs naturally, so people are a little more susceptible to ammonia. Even if exposed to ammonia, naturally, it would no effect due to such a small amount. A few traces of ammonia can be found in the air, your water, and even in your pets. Exposure to a high concentrate of ammonia can result in the following areas: skin, eyes, throat, and lungs. These areas are susceptible to minor burning and irritation. Ammonia can even cause death on severe occasions when there is a high level of exposure from this irritating substance.
I you happen to drink a little bit of ammonia, in concentrated solution form, it may burn your throat, mouth, and/or stomach. Getting some in your eyes could cause aggravating pain or, in some cases, even blindness.[3]
Uses
Ammonia has many uses such as Universal Indicator, Fertilizer, Refrigeration, Disinfectant, and Fuel, but the most important use of ammonia would have to be how well it produces nitric acid. Nitric acid is produced through the oxidation of ammonia. This is an important catalyst and contributes greatly to the production of fertilizer and explosives. Ammonia is also an indicator of many things, one example is how well it can detect if gas was present in a certain area.
Not only is it an ingredient in actual fertilizer, but it is also a mini-fertilizer in itself. Due to ammonia's great thermal properties, it was used as the main refrigerator unit, until dichlorodifluoromethane was discovered. Ammonia was replaced because of its toxic and corrosive behavior.
Ammonia is known better as a cleaning agent, or disinfectant. Often, ammonia will be combined with drinking water and chlorine to form a common disinfectant.[4]
 Browse |
Gallery
Related References
See Also
| ||||||||||||||