Dioxin
| Dioxin | |
|---|---|
| |
| General | |
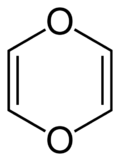
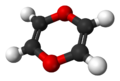
| Systematic name | 1,4-dioxin |
| Other names | p-dioxin or dioxin |
| Empirical formula | C4H4O2 |
| SMILES | O1C2=C(C=CC=C2)OC3=CC=CC=C13 |
| Molar mass | Molar mass::84.07 g/mol |
| Appearance | colorless liquid |
| CAS number | CAS number::290-67-5 |
| Properties | |
| Density and phase | [[Density::1.83 g/cm3]] liquid |
| Solubility | 13 x 10-9 g/l |
| Boiling point | Boiling point::75°C |
| Hazards | |
| Main hazards | toxic, very flammable |
| Related compounds | |
| Related compounds |
|
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Disclaimer and references | |
Dioxins are organic compounds that are not produced intentionally but only as a product of other chemical procedures such as incineration, wood and fuel burning, combustion, and others. When Dioxins enter the food chain, humans retain substances of it in their fatty tissue. Though dioxins can be reduced they are very difficult to remove completely from a certain location because they are resistant to harmful chemicals. Dioxins can be broken down slowly in sunlight. They are chemically classified as halogenated aromatic hydrocarbons. They are structurally comprised of two benzene rings joined by oxygen atoms, and they include these congeners: 10 dichlorodibenzo-p-dioxins (DCDD), 14 trichlorodibenzo-p-dioxins (TrCDD), and 22 tetrachlorodibenzo-p-dioxins (TCDD). [1]
Occurrences
Dioxins are used only for experimentation. For this reason, large amounts of dioxin have been exposed to the environment unintentionally as by-products of other chemical processes. As a result, they were banned from using commercially. Paper manufacturers that use chlorine dioxide in their processes brought dioxin to surface water, but since then, guidelines have been put into motion to stop and reduce the production of dioxin now and later in the future. Other contributors to dioxins' involvement in the environment include medical/hazardous waste, sewage sludge incinerators, diesel fuel combustion, and the burning of wood and residential oil. Dioxin can also be found in natural fires. Through incineration and combustion, huge levels of dioxin disperse throughout the atmosphere. This action causes dioxin to pervade into soil and sediments in water. [2]
Uses
Dioxin is used only in chemical research. It has no beneficial use to the natural world. It is a by-product of other chemical processes. Examples of some chemical processes are that of 2,4,5-trichlorophenoxyacetic acid and hexachlorophene. 2,4,5-trichlorophenoxyacetic acid was once used as a weed killer and hexachlorophene was used in cleaning products. Both of these compounds have long been banned from use in the United States. [3]
Properties
Dioxin is a liquid with no unique or identifying color. Its chemical formula is C4H4O2 and has a molecular mass of 84.07 g/mol. The boiling point for Dioxin is at 75°C or at 348°K. It is a highly flammable compound. [4]
| 2,3,7,8 TCDD |
|---|

|

|
2,3,7,8-Tetrachlorodibenzo-p-Dioxin, commonly called TCDD, is the most toxic type of Dioxin. It is used to measure the toxicity of other chemicals and compounds. Aside from research, TCDD is not made deliberately in the environment. [5] It is a odorless and colorless solid and has a molecular weight of 322 g/mol. Its chemical formula is C12H4Cl4O2. Low amounts of TCDD can be found in soils, water, and food and can be a source of exposure. Through TCDD, populations suffer from a number of illnesses such as birth defects, cancer risks, and chloracne. [6]
Effects
During the war in Vietnam, there was a plant killer that was called Agent Orange. It was made to get rid of plants and other vegetation growing in Vietnam in order to see clearer on the battlefield. They sprayed the chemicals into the air, not knowing the results that would follow. Soon the vegetation stopped growing, but along with animal sicknesses, humans were also greatly affected by it. Unusual cases started sprouting up and many people were seriously damaged. Even today, Dioxin has been present in our society, by our desire or not. [7]
Health and Environmental Effects
Serious health effects of Dioxin to humans can occur in several ways. In some cases of short-term and long-term exposure to Dioxin, humans have been known to display signs of extreme chloracne, which is an acne-like condition that develops within months of the first intake of Dioxin. Some illnesses connected to inhalation of the levels of Dioxin are lung cancer, soft-tissue sarcomas, lymphomas, and stomach carcinomas. [8]
Various amounts of Dioxin have a large effect on animals as well as humans. Animals such as dogs, monkeys, and guinea pigs show dangerous side effects to 2,3,7,8-TCDD, a type of Dioxin. Animal reactions to long-term exposure from this compound include hair loss, a decrease in body weight, and a weak immune system. Looking at animals' development and reproductive testing, we see that forms of skeletal deformities, kidney defects, altered levels of sex hormones and other several cases have become evident. [9]
TCDD can be transported and carried by the air, water, sediments and soil. When it is dispersed into the air, it can travel a long ways, even worldwide. Once it reaches animals, the compound can work its way into the food chain, ultimately affecting humans as well. Some traces of dioxin can be found in some of the population even today. [10] The largest source of dioxin surrounding us is our food. This is because Dioxin is fat-soluble, thus it easily makes its way up the food chain. North Americans receive 93% of Dioxin in meat and dairy products alone in a typical diet. According to a study done in May of 2001, the best way to keep away from Dioxin is to take on either a vegan outlook or vegan diet. Men and women have their own ways of relieving themselves of the Dioxin n their systems. Men only have the option of letting it gradually break down on its own, but women who are pregnant get rid of it through the placenta, straight into the growing baby or through their breast milk. [11]
References
- Dioxins Ces.iisc.ernet.in, Environmental Handbook, German Federal Ministry for Economic Cooperation and Development Bundesministerium für wirtschaftliche Zusammenarbeit und Entwicklung (BMZ)
- Dioxin Science Clarified, Advameg Inc.
- 2,3,7,8-Tetrachlorodibenzo-p-Dioxin (2,3,7,8,-TCDD) Technology Transfer Network Air Toxics Web Site, EPA.gov
- Polychlorinated dioxins and furans fact sheet Austrailian Government: Department of the Environment, Water, Heritage and the Arts
- Environmental Occurrence EPA, Clu-In
- Polychlorinated dioxins and furans Austrailian Government: Department of the Environment, Water, Heritage and the Arts
- Agent Orange Vietnam Veterans Ventura County, L. Vancil
- Dioxin (Chemical) Wikipedia.org
- Dioxin homepage Ejnet.org
| ||||||||||||||



