Isochron dating
Isochron dating is a common radiometric dating technique applied to date natural events like the crystallization of minerals as they cool, changes in rocks by metamorphism, or what are essentially naturally occurring shock events like meteor strikes. Minerals present in these events contain various radioactive elements which decay and the resulting daughter elements can then be used to deduce the age of the mineral through an isochron. The appeal of isochron dating is that it does not presuppose the initial amount of the daughter nuclide in the decay sequence. Indeed, the initial amount is not important because it can be found through this type of dating. [1]
The Problems and Process
Isochron dating began when scientists recognized difficulties with the assumptions of radiometric dating, especially how much of the daughter products might have been present when the mineral first formed. Isochron dating has been developed in an attempt to solve such problems.
| PI | Parent (radioactive) isotope |
| DI | Non-radiogenic isotope of same element as daughter isotope |
| DE | Daughter isotope |
According to theory, the sample starts out with daughter isotopes present at constant ratios in relation to one another, but with the parent isotope the ratio is arbitrary. As a result it can be displayed in the form of a straight horizontal line on a graph. As the parent decays to daughter the ratios change and the straight line remains but becomes angled. The slope of the line equals the number of half-lives the parent isotope has passed since solidification.
If there occurs a gain or loss of parent isotope the point moves horizontally. If there is a gain then the point moves right and finally if it is a loss the point moves left.
If there is a gain or loss of daughter isotope the point moves vertically. A gain moves the point up and a loss moves the point down.
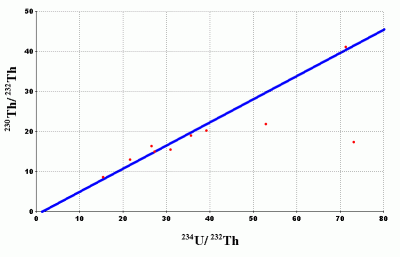
For example, take this set of data points.
A shift from contamination can take place in all of the data points, but such contamination does not affect all data points equally, so it can cause the data points to shift off the true isochron completely. Given this, when one looks at an isochron plot, how can one really tell where the true isochron line should be? Sufficient contamination can produce any isochron pattern regardless of the true isochron.
It is even possible to get a negative slope. This would be equivalent to a negative or future date. Potassium-Argon
When you look at actual isochron plots such as the ones at the above source, there seems to be room for subjectivity. Some are better than others but there is often room for multiple plot lines. Even uniformitarian geologists recognize the existence of false isochrons, so how do they distinguish good data from bad? The answer is where the sample fits in the Geologic Column assumption quite prevalent within evolutionary science today. [2]
An Example of A Real Isochron
Reconstruction of an isochron from original data obtained from the Peat Deposits From the Site Fili Park. [3] This isochron of the Fili Park Peat deposits indicates an age of 89,000 years. This is the isochron presented by the author of the paper cited above. It is a fairly good five point isochron.
It is also supported by additional isotope ratios. In addition the data points were the five out of ten samples that, "were interpreted to be in the more or less closed-system." However it was concluded that this isochron was the result of contamination as an open system based on the fact that the date was about 50% of the age suggested by the spore and pollen fossils in the deposit. So this otherwise good isochron was rejected because it disagreed with the fossils or essentially the pre-determined view of where those fossils are buried within the Geologic column.
Even when the samples that were originally considered part of an open system are added the result is still a fairly good isochron with only two points showing significant deviation.
If the two deviating points are removed the result is an excellent eight point isochron.
Furthermore, all ten data points form a fairly good second isotope ratio plot.
Conclusion
The result is that contamination can form good looking isochron data and uniformitarian geologists know it. The real way a "true" isochron is distinguished from a false isochron is by how well it agrees with how old the fossils are considered to be.
References
- Isochron Dating by Answers
- New Age Data of Buried Peat Deposits From the Site "Fili Park" by the Journal on Methods and Applications of Absolute Chronology
- Basics of Radioactive Isotope Geochemistry by Cornell University
| ||||||||||||||||||||