Glucose
| Glucose | |
|---|---|
| |
| General | |
| Systematic name | Pentahydroxyhexanal |
| Other names |
Blood sugar |
| Molecular formula | C6H12O6 |
| SMILES |
OC[C@H]1OC(O)[C@H](O) |
| Molar mass | Molar mass::180.16 g/mol |
| Appearance | White powder |
| CAS number | CAS number::50-99-7 |
| Properties | |
| Density and phase | Density::1.54 g/ml |
| Solubility in water | 90.8 g/100 ml (25°C) |
| Melting point | Melting point::146°C |
| Boiling point | Boiling point::150°C |
| Structure | |
| Crystal structure | Hexagonal |
| Dipole moment | 1.8 D |
| Hazards | |
| MSDS | Material Safety Data Sheet |
| Main hazards | Flammable |
| NFPA 704 | |
| R/S statement | R: R10 S: S9, S17 |
| RTECS number | LZ6600000 |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Disclaimer and references | |
Glucose is a simple sugar known as a monosaccharide. This compound is essential to life and is the main energy carrier used by almost all living organisms. This sugar is taken into the cells and transformed by the mitochondria into ATP during a complex reaction called cellular respiration. This simple sugar can be found in almost every edible substance, and in large quantity in saps of plants from [[photosynthesis]. Many of our foods contain glucose, while we think that only sweets and bread, vegetables and fruits do as well.
Glucose levels in the body are controlled by the hormones insulin and glucagon. When the pancreas does not produce the right amounts of insulin, diabetes may be contracted.
Properties
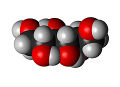
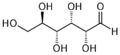
Glucose is known as the most important sugar in science. A monosaccharide, or simple sugar, can be known by the names D-glucose and also dextrose. Glucose holds six carbon atoms and may form many kinds of structures as it could either occur in an open chain or a ring form. [1] Glucose molecules are often made up of many single bond and one double bonds. There is also the L-glucose molecule, that is much more rare compared to the D-glucose molecule. D-glucose is what animals and humans use to create energy. L-glucose can never be used by your body to create energy, however. [2]
Both molecules of glucose have the same formula and structure. All glucose is white and powdery, the look of sugar. The general molecular weight is 180.16 grams per mole. Its melting point ranges between 153-156 degrees Celsius, or 307-312 degrees Fahrenheit. The only dangers of glucose is that it is flammable. It may react catastrophically with substances such as potassium nitrate and sodium peroxide. However, this compound will always remain stable under 50 degrees Celsius. It is well known that most foods we eat are converted to glucose to be used for energy. With this information, it is understandable that only one mole of glucose holds 2870 kilo-joules of energy! [3]
Synthesis / Occurrences
Glucose is one of the most simple sugars. It is the most essential in every human metabolism. Because it is a simple unit of sugar, it is named a monosaccharide. This molecule is a carbohydrate, and is one of the smallest carbohydrates in its class. People may refer to glucose as "blood sugar" because it is found in the human bloodstream. This is the sugar that gives energy to people, and allows plants to produce food. It is used to produce water, carbon dioxide, and nitrogen when in the human body. When a plant goes through photosynthesis, it utilizes the sun to create glucose molecules. This is what we are able to find in the sap of plants, which we use to create corn syrup. [4]
Because the human body relies so highly on glucose, it may produce it without help. This molecule is both an energy used by the brain and the red blood cells. Without it, our bodies would surely fail. Our active human bodies need a supplement of 160g of glucose each day to function properly. [5] Glucose is the base in our bodies by which we make fats, carboxylic acids, and amino acids. It is synthesized by the combination of pyruvic acid and acetyl CoA. [6]
Uses
Glucose is used in the body to produce energy for reactions performed by cells of the body. Glucose is also used in plants as a food source when produced through photosynthesis. Plants also use it for energy so they can release oxygen and take in carbon dioxide. It can be found in the many carbohydrates that we eat. This compound can be used to treat hypoglycemia in people. Hypoglycemia is a lack of sugar in the blood. A person is considered to have low blood sugar when there is 70 mL per deciliter in the body. People with diabetes could very well develop hypoglycemia and it can be fatal. Those with this illness are advised to take either supplements of the compound or drink sugary liquids to raise their blood sugar levels quickly and safely.
Glucose also has good uses for athletes. High-endurance athletes must have an efficient way to refuel their blood with electrolytes. Drinks such as Gatorade that contain glucose may do this more efficiently that others because glucose absorbs into the bloodstream much more quickly than other sugars. The sweet taste of this compound also makes it a very common ingredient in many of the foods we consume. It is used in many sweets and candies, and can be an effective supplement for those attempting to gain weight. While giving a palatable taste to many foods, it also provides a source of energy to those who eat or drink it. A lack of glucose can be dangerous for any person, if it is not regularly consumed, but it is found in most foods. [7]
Insulin and Diabetes
Diabetes is the main disease that deals with your body's failure to properly utilize glucose. Diabetes is the state of having too much glucose in your bloodstream. This disease can be caused by many different things. One main reason someone may have diabetes is that their body does not produce the sufficient amounts of insulin. Insulin is what transports glucose to be turned into energy. With little insulin, excessive amounts of sugar will not be changed into usable energy. People are diagnosed with diabetes when their blood glucose levels are much higher than average. The effects of diabetes may include blindness, and diseases in the kidneys or heart. [8]
Doctors have come up with an insulin shot that diabetes patients may take in order to live more regular and safe lives. Insulin therapy may be one of the most important steps of diabetes treatment. Because insulin plays such an important role in regulating your blood sugar, patients must take regular doses or shots in order to keep their levels under control. The pancreas in the human body is what normally creates insulin. If the pancreas is not at a healthy level, little to no insulin may be produced. When insulin does not do its job, glucose is unable to enter into your body's cells. This shuts off your energy resources and can be very dangerous. With insulin shots, patients with type 1 diabetes and type 2 diabetes may be able to convert the sugars in their bodies to energy properly. [9]
Video
The Glucose song!
References
- ↑ Glucose Edinformatics. Web. 22 January 2014 (Date-Accessed).
- ↑ Physical Properties of Glucose eHow. Web. 22 January 2014 (Date-Accessed).
- ↑ What Are The Properties of Glucose eHow. Web. 22 January 2014 (Date-Accessed).
- ↑ Nave, R. Glucose HyperPhysics. Web. 8 January 2014 (Date-Accessed).
- ↑ Glucose can be Synthesized. . . NCBI. Web. 8 January 2014 (Date-Accessed).
- ↑ Glucose Clackamas. Web. 8 January 2014 (Date-Accessed).
- ↑ Stein, Natalie. Uses of Glucose Livestrong. Web. 16 August 2013 (Date-Published).
- ↑ Diabetes: Definition, Causes, and Symptoms Davita. Web. 22 January 2014 (Date-Accessed).
- ↑ Diabetes Treatment MayoClinic. Web. 22 January 2014 (Date-Accessed).
| ||||||||||||||




